Donepezil: Perbedaan antara revisi
WanaraLima (bicara | kontrib) Dibuat dengan menerjemahkan halaman "User:Mr. Ibrahem/Donepezil" |
+ 7 Kategori; ± 2 Kategori menggunakan HotCat |
||
| (Satu revisi perantara oleh satu pengguna lainnya tidak ditampilkan) | |||
| Baris 3: | Baris 3: | ||
<!-- Names -->|tradename=Aricept, others|synonyms=|IUPAC_name=(''RS'')-2-[(1-Benzyl-4-piperidyl)methyl]-5,6-dimethoxy-2,3-dihydroinden-1-one |
<!-- Names -->|tradename=Aricept, others|synonyms=|IUPAC_name=(''RS'')-2-[(1-Benzyl-4-piperidyl)methyl]-5,6-dimethoxy-2,3-dihydroinden-1-one |
||
<!-- Clinical data -->|pregnancy_AU=B3|pregnancy_US=C|routes_of_administration=By mouth ([[Tablet (pharmacy)|tablets]]) Absorption is not affected by food or time of day.<ref name=Kum2020 |
<!-- Clinical data -->|pregnancy_AU=B3|pregnancy_US=C|routes_of_administration=By mouth ([[Tablet (pharmacy)|tablets]]) Absorption is not affected by food or time of day.<ref name=Kum2020 />|Drugs.com={{drugs.com|monograph|donepezil-hydrochloride}}|MedlinePlus=a697032 |
||
<!-- Legal data -->|legal_AU=S4|legal_US=Rx-only|legal_status=Rx-only|DailyMedID=Donepezil |
<!-- Legal data -->|legal_AU=S4|legal_US=Rx-only|legal_status=Rx-only|DailyMedID=Donepezil |
||
<!-- Pharmacokinetic data -->|bioavailability=100%<ref name=Kum2020 |
<!-- Pharmacokinetic data -->|bioavailability=100%<ref name=Kum2020 /><ref name="Seltzer 2005 pp. 527–536">{{cite journal | last=Seltzer | first=Ben | title=Donepezil: a review | journal=Expert Opinion on Drug Metabolism & Toxicology | publisher=Informa Healthcare | volume=1 | issue=3 | date=2005-09-29 | issn=1742-5255 | pmid=16863459 | doi=10.1517/17425255.1.3.527 | pages=527–36 | quote=there is a linear relationship between dose and pharmacodynamic effects, measured as red blood cell acetylcholinesterase inhibition and clinical efficacy. Despite being 96% bound to plasma proteins, it has few interactions with other drugs, and the 5-mg dose can be given safely to patients with mild-to-moderate hepatic and renal-disease. }}</ref>|protein_bound=96%, [[albumin]] (about 75%) and [[alpha1-acid glycoprotein]] (21%).<ref name="Seltzer 2005 pp. 527–536" /><ref name=Kum2020 />|metabolism=[[CYP2D6]], [[CYP3A4]], and [[glucuronidation]].<ref name=Kum2020 /> Four major metabolites, two of which are active.<ref name="Seltzer 2005 pp. 527–536" /><ref name=Kum2020 />|elimination_half-life=70 hours<ref name="Asiri Mostafa 2010 pp. 117–150">{{cite book | last1=Asiri | first1=Yousif A. | last2=Mostafa | first2=Gamal A.E. | title=Profiles of Drug Substances, Excipients and Related Methodology | chapter=Donepezil | publisher=Elsevier | year=2010 | volume=35 | isbn=978-0-12-380884-4 | issn=1871-5125 | doi=10.1016/s1871-5125(10)35003-5 | pages=117–50 | pmid=22469221 | quote=Plasma donepezil concentrations decline with a half-life of approximately 70 h. Sex, race, and smoking history have no clinically significant influence on plasma concentrations of donepezil [46–51].}}</ref> Around 100 hours in elderly patients.<ref name=Kum2020 />|excretion=0.11–0.13 (L/h/kg); excreted mostly by the kidneys. Around 17% is excreted unchanged in the urine. About 15% to 20% is excreted in feces.<ref name=Kum2020 /><ref name="Seltzer 2005 pp. 527–536" /> Steady-state clearance is similar at all ages.<ref name=Kum2020 /> |
||
<!-- Chemical and physical data -->|C=24|H=29|N=1|O=3|smiles=O=C2c1cc(OC)c(OC)cc1CC2CC4CCN(Cc3ccccc3)CC4|StdInChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3|StdInChI_Ref={{stdinchicite|correct|chemspider}}|StdInChIKey=ADEBPBSSDYVVLD-UHFFFAOYSA-N|StdInChIKey_Ref={{stdinchicite|correct|chemspider}}}} |
<!-- Chemical and physical data -->|C=24|H=29|N=1|O=3|smiles=O=C2c1cc(OC)c(OC)cc1CC2CC4CCN(Cc3ccccc3)CC4|StdInChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3|StdInChI_Ref={{stdinchicite|correct|chemspider}}|StdInChIKey=ADEBPBSSDYVVLD-UHFFFAOYSA-N|StdInChIKey_Ref={{stdinchicite|correct|chemspider}}}} |
||
'''Donepezil''' adalah obat yang digunakan untuk mengatasi[[penyakit Alzheimer]] . <ref name="AHFS2019">{{Cite web|title=Donepezil Hydrochloride Monograph for Professionals|url=https://www.drugs.com/monograph/donepezil-hydrochloride.html|website=Drugs.com|publisher=American Society of Health-System Pharmacists|language=en|archive-url=https://web.archive.org/web/20190726092537/https://www.drugs.com/monograph/donepezil-hydrochloride.html|archive-date=26 July 2019|access-date=4 February 2019|url-status=live}}</ref> Donepezil bisa memberikan cukup manfaat untuk fungsi mental. <ref>{{Cite journal|date=June 2018|title=Donepezil for dementia due to Alzheimer's disease|journal=The Cochrane Database of Systematic Reviews|volume=6|pages=CD001190|doi=10.1002/14651858.CD001190.pub3|pmc=6513124|pmid=29923184|vauthors=Birks JS, Harvey RJ}}</ref> Sayangnya penggunaan obat ini masih belum terbukti dalam mengubah penyakit alzheimer. <ref>{{Cite journal|last=Swedish Council on Health Technology Assessment|date=June 2008|title=Dementia – Caring, Ethics, Ethnical and Economical Aspects: A Systematic Review|pmid=28876770}}</ref> Penggunaan harus dihentikan jika tidak memiliki manfaat atau perubahan yang terlihat. <ref name="BNF76">{{Cite book|date=2018|title=British national formulary : BNF 76|publisher=Pharmaceutical Press|isbn=9780857113382|edition=76|pages=300|ref={{harvid | BNF76 |2018 }}}}</ref> Rute pemberian obat ini melalui oral. <ref name="AHFS2019" |
'''Donepezil''' adalah obat yang digunakan untuk mengatasi[[penyakit Alzheimer]] . <ref name="AHFS2019">{{Cite web|title=Donepezil Hydrochloride Monograph for Professionals|url=https://www.drugs.com/monograph/donepezil-hydrochloride.html|website=Drugs.com|publisher=American Society of Health-System Pharmacists|language=en|archive-url=https://web.archive.org/web/20190726092537/https://www.drugs.com/monograph/donepezil-hydrochloride.html|archive-date=26 July 2019|access-date=4 February 2019|url-status=live}}</ref> Donepezil bisa memberikan cukup manfaat untuk fungsi mental. <ref>{{Cite journal|date=June 2018|title=Donepezil for dementia due to Alzheimer's disease|journal=The Cochrane Database of Systematic Reviews|volume=6|pages=CD001190|doi=10.1002/14651858.CD001190.pub3|pmc=6513124|pmid=29923184|vauthors=Birks JS, Harvey RJ}}</ref> Sayangnya penggunaan obat ini masih belum terbukti dalam mengubah penyakit alzheimer. <ref>{{Cite journal|last=Swedish Council on Health Technology Assessment|date=June 2008|title=Dementia – Caring, Ethics, Ethnical and Economical Aspects: A Systematic Review|pmid=28876770}}</ref> Penggunaan harus dihentikan jika tidak memiliki manfaat atau perubahan yang terlihat. <ref name="BNF76">{{Cite book|date=2018|title=British national formulary : BNF 76|publisher=Pharmaceutical Press|isbn=9780857113382|edition=76|pages=300|ref={{harvid | BNF76 |2018 }}}}</ref> Rute pemberian obat ini melalui oral. <ref name="AHFS2019" /> |
||
Kejadian efek samping yang umum pada penggunaan obat ini seperti , rasa lelah,sulit tidur, agresi, diare, mual dan kram otot. <ref name="AHFS2019" |
Kejadian efek samping yang umum pada penggunaan obat ini seperti , rasa lelah,sulit tidur, agresi, diare, mual dan kram otot. <ref name="AHFS2019" /> <ref name="BNF76" /> Kejadian efek samping yang serius pada penggunaan obat ini meliputi [[aritmia]], [[retensi urin]], dan [[sawan]] . <ref name="AHFS2019" /> Obati ini merupakan golongan inhibitor asetilkolinesterase reversibel yang memiliki mekanisme kerja [[Sistem saraf pusat|secara terpusat]] dan secara struktural tidak berkaitan dengan agen antikolinesterase lainnya. <ref name="AHFS2019" /> <ref name=Kum2020>{{citation | last1=Kumar | first1=A | last2=Sharma | first2=S | title=Donepezil | chapter=article-20656 | publisher=StatPearls Publishing | location=Treasure Island (FL) | year=2020 | pmid=30020629 | url=http://www.ncbi.nlm.nih.gov/books/NBK513257/ | access-date=2020-04-12 | archive-date=2021-08-28 | archive-url=https://web.archive.org/web/20210828064644/https://www.ncbi.nlm.nih.gov/books/NBK513257/ | url-status=live }}</ref> |
||
Obat donepezil disetujui dalam penggunaan medis di Amerika Serikat pada tahun 1996. <ref name="AHFS2019" /> Donepezil sudah tersedia dalam bentuk [[obat generik]]. <ref name="BNF76" /> |
|||
Obat donepezil disetujui dalam penggunaan medis di Amerika Serikat pada tahun 1996. <ref name="AHFS2019">{{Cite web|title=Donepezil Hydrochloride Monograph for Professionals|url=https://www.drugs.com/monograph/donepezil-hydrochloride.html|website=Drugs.com|publisher=American Society of Health-System Pharmacists|language=en|archive-url=https://web.archive.org/web/20190726092537/https://www.drugs.com/monograph/donepezil-hydrochloride.html|archive-date=26 July 2019|access-date=4 February 2019|url-status=live}}<cite class="citation web cs1" data-ve-ignore="true">[https://www.drugs.com/monograph/donepezil-hydrochloride.html "Donepezil Hydrochloride Monograph for Professionals"]. </cite></ref> Donepezil sudah tersedia dalam bentuk [[obat generik]]. <ref name="BNF76">{{Cite book|date=2018|title=British national formulary : BNF 76|publisher=Pharmaceutical Press|isbn=9780857113382|edition=76|pages=300|ref={{harvid | BNF76 |2018 }}}}<cite class="citation book cs1" data-ve-ignore="true" id="CITEREFBNF762018">''British national formulary : BNF 76'' (76 ed.). </cite></ref> |
|||
== Referensi == |
== Referensi == |
||
[[Kategori: |
[[Kategori:Penghambat asetilkolinesterase]] |
||
[[Kategori: |
[[Kategori:Obat antidemensia]] |
||
[[Kategori:Pengobatan penyakit Alzheimer]] |
|||
[[Kategori:Nootropika]] |
|||
[[Kategori:Piperidina]] |
|||
[[Kategori:Eter katekol]] |
|||
[[Kategori:Indana]] |
|||
[[Kategori:Agonis sigma]] |
|||
[[Kategori:Obat yang dikembangkan oleh Pfizer]] |
|||
Revisi per 22 Mei 2024 20.37
| |
|---|---|
| Nama sistematis (IUPAC) | |
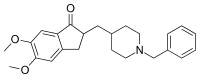
| (RS)-2-[(1-Benzyl-4-piperidyl)methyl]-5,6-dimethoxy-2,3-dihydroinden-1-one | |
| Data klinis | |
| Nama dagang | Aricept, others |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a697032 |
| Data lisensi | US Daily Med:pranala |
| Kat. kehamilan | B3(AU) C(US) |
| Status hukum | Harus dengan resep dokter (S4) (AU) ℞-only (US) ℞ Preskripsi saja |
| Rute | By mouth (tablets) Absorption is not affected by food or time of day.[1] |
| Data farmakokinetik | |
| Bioavailabilitas | 100%[1][2] |
| Ikatan protein | 96%, albumin (about 75%) and alpha1-acid glycoprotein (21%).[2][1] |
| Metabolisme | CYP2D6, CYP3A4, and glucuronidation.[1] Four major metabolites, two of which are active.[2][1] |
| Waktu paruh | 70 hours[3] Around 100 hours in elderly patients.[1] |
| Ekskresi | 0.11–0.13 (L/h/kg); excreted mostly by the kidneys. Around 17% is excreted unchanged in the urine. About 15% to 20% is excreted in feces.[1][2] Steady-state clearance is similar at all ages.[1] |
| Pengenal | |
| Kode ATC | ? |
| Data kimia | |
| Rumus | C24H29NO3 |
| SMILES | eMolecules & PubChem |
| |
Donepezil adalah obat yang digunakan untuk mengatasipenyakit Alzheimer . [4] Donepezil bisa memberikan cukup manfaat untuk fungsi mental. [5] Sayangnya penggunaan obat ini masih belum terbukti dalam mengubah penyakit alzheimer. [6] Penggunaan harus dihentikan jika tidak memiliki manfaat atau perubahan yang terlihat. [7] Rute pemberian obat ini melalui oral. [4]
Kejadian efek samping yang umum pada penggunaan obat ini seperti , rasa lelah,sulit tidur, agresi, diare, mual dan kram otot. [4] [7] Kejadian efek samping yang serius pada penggunaan obat ini meliputi aritmia, retensi urin, dan sawan . [4] Obati ini merupakan golongan inhibitor asetilkolinesterase reversibel yang memiliki mekanisme kerja secara terpusat dan secara struktural tidak berkaitan dengan agen antikolinesterase lainnya. [4] [1]
Obat donepezil disetujui dalam penggunaan medis di Amerika Serikat pada tahun 1996. [4] Donepezil sudah tersedia dalam bentuk obat generik. [7]
Referensi
- ^ a b c d e f g h i Kumar, A; Sharma, S (2020), "article-20656", Donepezil, Treasure Island (FL): StatPearls Publishing, PMID 30020629, diarsipkan dari versi asli tanggal 2021-08-28, diakses tanggal 2020-04-12
- ^ a b c d Seltzer, Ben (2005-09-29). "Donepezil: a review". Expert Opinion on Drug Metabolism & Toxicology. Informa Healthcare. 1 (3): 527–36. doi:10.1517/17425255.1.3.527. ISSN 1742-5255. PMID 16863459.
there is a linear relationship between dose and pharmacodynamic effects, measured as red blood cell acetylcholinesterase inhibition and clinical efficacy. Despite being 96% bound to plasma proteins, it has few interactions with other drugs, and the 5-mg dose can be given safely to patients with mild-to-moderate hepatic and renal-disease.
- ^ Asiri, Yousif A.; Mostafa, Gamal A.E. (2010). "Donepezil". Profiles of Drug Substances, Excipients and Related Methodology. 35. Elsevier. hlm. 117–50. doi:10.1016/s1871-5125(10)35003-5. ISBN 978-0-12-380884-4. ISSN 1871-5125. PMID 22469221.
Plasma donepezil concentrations decline with a half-life of approximately 70 h. Sex, race, and smoking history have no clinically significant influence on plasma concentrations of donepezil [46–51].
- ^ a b c d e f "Donepezil Hydrochloride Monograph for Professionals". Drugs.com (dalam bahasa Inggris). American Society of Health-System Pharmacists. Diarsipkan dari versi asli tanggal 26 July 2019. Diakses tanggal 4 February 2019.
- ^ Birks JS, Harvey RJ (June 2018). "Donepezil for dementia due to Alzheimer's disease". The Cochrane Database of Systematic Reviews. 6: CD001190. doi:10.1002/14651858.CD001190.pub3. PMC 6513124
 . PMID 29923184.
. PMID 29923184.
- ^ Swedish Council on Health Technology Assessment (June 2008). "Dementia – Caring, Ethics, Ethnical and Economical Aspects: A Systematic Review". PMID 28876770.
- ^ a b c British national formulary : BNF 76 (edisi ke-76). Pharmaceutical Press. 2018. hlm. 300. ISBN 9780857113382.
