Aripiprazol: Perbedaan antara revisi
Tampilan
Konten dihapus Konten ditambahkan
k Bot: Perubahan kosmetika |
Rescuing 1 sources and tagging 0 as dead.) #IABot (v2.0.8 |
||
| Baris 26: | Baris 26: | ||
<!--Pharmacokinetic data--> |
<!--Pharmacokinetic data--> |
||
| bioavailability = 87%<ref name = ABILIFY>{{cite web|title=Product Information for ABILIFY<sup>TM</sup> Aripiprazole Tablets & Orally Disintegrating Tablets|work = TGA eBusiness Services|publisher = Bristol-Myers Squibb Australia Pty Ltd|date=1 November 2012|accessdate=22 October 2013|url=https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2010-PI-03820-3}}</ref> |
| bioavailability = 87%<ref name = ABILIFY>{{cite web|title=Product Information for ABILIFY<sup>TM</sup> Aripiprazole Tablets & Orally Disintegrating Tablets|work = TGA eBusiness Services|publisher = Bristol-Myers Squibb Australia Pty Ltd|date=1 November 2012|accessdate=22 October 2013|url=https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2010-PI-03820-3}}</ref> |
||
<ref name = DM>{{cite web|title=ABILIFY (aripiprazole) tablet ABILIFY (aripiprazole) solution ABILIFY DISCMELT (aripiprazole) tablet, orally disintegrating ABILIFY (aripiprazole) injection, solution [Otsuka America Pharmaceutical, Inc.]|date=April 2013|accessdate=22 October 2013|url=http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=c040bd1d-45b7-49f2-93ea-aed7220b30ac|publisher=Otsuka America Pharmaceutical, Inc.|work=DailyMed}}</ref><ref name = EMC>{{cite web|title=Abilify Tablets, Orodispersible Tablets, Oral Solution - Summary of Product Characteristics (SPC)|date=20 September 2013|accessdate=22 October 2013|publisher=Otsuka Pharmaceuticals (UK) Ltd|work=electronic Medicines Compendium|url=http://www.medicines.org.uk/emc/medicine/18494/SPC/Abilify+Tablets%2c+Orodispersible+Tablets%2c+Oral+Solution/}}</ref><ref name = EMA>{{cite web|title=ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS|work=European Medicines Agency|publisher=Otsuka Pharmaceutical Europe Ltd.|accessdate=22 October 2013|url=http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000471/WC500020170.pdf}}</ref> |
<ref name = DM>{{cite web|title=ABILIFY (aripiprazole) tablet ABILIFY (aripiprazole) solution ABILIFY DISCMELT (aripiprazole) tablet, orally disintegrating ABILIFY (aripiprazole) injection, solution [Otsuka America Pharmaceutical, Inc.]|date=April 2013|accessdate=22 October 2013|url=http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=c040bd1d-45b7-49f2-93ea-aed7220b30ac|publisher=Otsuka America Pharmaceutical, Inc.|work=DailyMed}}</ref><ref name = EMC>{{cite web|title=Abilify Tablets, Orodispersible Tablets, Oral Solution - Summary of Product Characteristics (SPC)|date=20 September 2013|accessdate=22 October 2013|publisher=Otsuka Pharmaceuticals (UK) Ltd|work=electronic Medicines Compendium|url=http://www.medicines.org.uk/emc/medicine/18494/SPC/Abilify+Tablets%2c+Orodispersible+Tablets%2c+Oral+Solution/|archive-date=2016-03-04|archive-url=https://web.archive.org/web/20160304001126/http://www.medicines.org.uk/emc/medicine/18494/SPC/Abilify+Tablets,+Orodispersible+Tablets,+Oral+Solution/|dead-url=yes}}</ref><ref name = EMA>{{cite web|title=ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS|work=European Medicines Agency|publisher=Otsuka Pharmaceutical Europe Ltd.|accessdate=22 October 2013|url=http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000471/WC500020170.pdf}}</ref> |
||
| protein_bound = >99%<ref name = ABILIFY/><ref name = DM/><ref name = EMC/><ref name = EMA/> |
| protein_bound = >99%<ref name = ABILIFY/><ref name = DM/><ref name = EMC/><ref name = EMA/> |
||
| metabolism = [[Hepatik]] (hati; terutama via [[CYP3A4]] dan [[CYP2D6]]<ref name = ABILIFY/><ref name = DM/><ref name = EMC/><ref name = EMA/>) |
| metabolism = [[Hepatik]] (hati; terutama via [[CYP3A4]] dan [[CYP2D6]]<ref name = ABILIFY/><ref name = DM/><ref name = EMC/><ref name = EMA/>) |
||
Revisi per 19 Agustus 2021 20.02
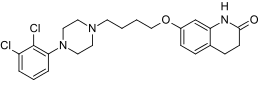
| |
|---|---|

| |
| Nama sistematis (IUPAC) | |
| 7-{4-[4-(2,3-Dichlorophenyl)piperazin-1-yl]butoxy}-3,4-dihydroquinolin-2(1H)-one | |
| Data klinis | |
| Nama dagang | Abilify |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a603012 |
| Data lisensi | EMA:Aripiprex pranala, US FDA:link |
| Kat. kehamilan | B3(AU) C(US) |
| Status hukum | Harus dengan resep dokter (S4) (AU) ℞-only (CA) POM (UK) ℞-only (US) |
| Rute | Oral (via tablet, tablet orodispersable, dan sirup); intramuskular (termasuk sebagai depot) |
| Data farmakokinetik | |
| Bioavailabilitas | 87%[1] |
| Ikatan protein | >99%[1][2][3][4] |
| Metabolisme | Hepatik (hati; terutama via CYP3A4 dan CYP2D6[1][2][3][4]) |
| Waktu paruh | 75 jam (metabolit aktif adalah 94 jam)[1][2][3][4] |
| Ekskresi | Ginjal (27%; <1% tidak berubah), Tinja (60%; 18% tidak berubah)[1][2][3][4] |
| Pengenal | |
| Nomor CAS | 129722-12-9 |
| Kode ATC | N05AX12 |
| PubChem | CID 60795 |
| Ligan IUPHAR | 34 |
| DrugBank | DB01238 |
| ChemSpider | 54790 |
| UNII | 82VFR53I78 |
| KEGG | D01164 |
| ChEBI | CHEBI:31236 |
| ChEMBL | CHEMBL1112 |
| Data kimia | |
| Rumus | C23H27Cl2N3O2 |
| Massa mol. | 448.385 g/mol |
| SMILES | eMolecules & PubChem |
| |
Aripiprazol, dijual dengan nama merek Abilify adalah antipsikotik atipikal. Obat dianjurkan dan terutama digunakan dalam pengobatan skizofrenia dan gangguan bipolar.[5] Kegunaan lain termasuk sebagai terapi tambahan pada penyakit depresi, gangguan tic, dan iritabilitas yang berhubungan dengan autisme.[6] Menurut review Cochrane, bukti bentuk oral pada skizofrenia tidak cukup untuk menentukan efek pada fungsi umum.[7] Selain itu, karena banyak orang putus obat selama percobaan sebelum mereka selesai, keseluruhan kekuatan kesimpulan adalah rendah.[7]
Referensi
- ^ a b c d e "Product Information for ABILIFYTM Aripiprazole Tablets & Orally Disintegrating Tablets". TGA eBusiness Services. Bristol-Myers Squibb Australia Pty Ltd. 1 November 2012. Diakses tanggal 22 October 2013.
- ^ a b c d e "ABILIFY (aripiprazole) tablet ABILIFY (aripiprazole) solution ABILIFY DISCMELT (aripiprazole) tablet, orally disintegrating ABILIFY (aripiprazole) injection, solution [Otsuka America Pharmaceutical, Inc.]". DailyMed. Otsuka America Pharmaceutical, Inc. April 2013. Diakses tanggal 22 October 2013.
- ^ a b c d e "Abilify Tablets, Orodispersible Tablets, Oral Solution - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Otsuka Pharmaceuticals (UK) Ltd. 20 September 2013. Diarsipkan dari versi asli tanggal 2016-03-04. Diakses tanggal 22 October 2013.
- ^ a b c d e "ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS" (PDF). European Medicines Agency. Otsuka Pharmaceutical Europe Ltd. Diakses tanggal 22 October 2013.
- ^ "abilify". The American Society of Health-System Pharmacists. Diakses tanggal 3 April 2011.
- ^ http://www.webmd.com/drugs/drug-64439-Abilify+Oral.aspx?drugid=64439&drugname=Abilify+Oral&source=1
- ^ a b Belgamwar RB, El-Sayeh HG (Aug 10, 2011). "Aripiprazole versus placebo for schizophrenia". The Cochrane database of systematic reviews (8): CD006622. doi:10.1002/14651858.CD006622.pub2. PMID 21833956.
