Pengguna:Agung.karjono/Bak pasir/Tembaga: Perbedaan antara revisi
←Membuat halaman berisi 'Tembaga' |
kTidak ada ringkasan suntingan |
||
| (16 revisi perantara oleh 4 pengguna tidak ditampilkan) | |||
| Baris 1: | Baris 1: | ||
{{Other uses}} |
|||
Tembaga |
|||
{{Use dmy dates|date=July 2014}} |
|||
{{Infobox copper}} |
|||
'''Copper''' is a [[chemical element]] with symbol '''Cu''' (from {{lang-la|cuprum}}) and [[atomic number]] 29. It is a soft, malleable, and [[ductility|ductile]] metal with very high [[thermal conductivity|thermal]] and [[electrical conductivity]]. A freshly exposed surface of pure copper has a reddish-orange color. Copper is used as a conductor of heat and electricity, as a building material, and as a constituent of various metal [[alloy]]s, such as [[sterling silver]] used in [[jewelry]], [[cupronickel]] used to make marine hardware and [[coins]], and [[constantan]] used in [[strain gauge]]s and [[thermocouples]] for temperature measurement. |
|||
Copper is one of the few metals that occur in nature in directly usable metallic form as opposed to needing extraction from an ore. This led to very early human use, from c. 8000 BC. It was the first metal to be [[Smelting|smelted]] from its ore, c. 5000 BC, the first metal to be cast into a shape in a mold, c. 4000 BC and the first metal to be purposefully alloyed with another metal, [[tin]], to create [[bronze]], c. 3500 BC.<ref name="EncBrit">{{cite book |editor1-last=McHenry |editor1-first=Charles |title=The New Encyclopedia Britannica |date=1992 |publisher=Encyclopedia Britannica, Inc. |location=Chicago |isbn=0-85229-553-7 |page=612 |volume=3 |edition=15}}</ref> |
|||
In the [[ancient Rome|Roman era]], copper was principally mined on [[Cyprus]], the origin of the name of the metal, from ''aes сyprium'' (metal of Cyprus), later corrupted to ''сuprum'', from which the words ''copper'' (English), ''cuivre'' (French), ''Koper'' (Dutch) and ''Kupfer'' (German) are all derived.<ref>''Encyclopaedia Britannica'', 11th ed., vol. 7, p. 102.</ref> The commonly encountered compounds are copper(II) salts, which often impart blue or green colors to such minerals as [[azurite]], [[malachite]], and [[turquoise]], and have been used widely and historically as pigments. Copper used in buildings, usually for roofing, oxidizes to form a green [[verdigris]] (or [[patina]]). Copper is sometimes used in [[decorative art]], both in its elemental metal form and in compounds as pigments. Copper compounds are used as [[bacteriostatic agent]]s, [[fungicide]]s, and wood preservatives. |
|||
Copper is essential to all living organisms as a trace [[dietary mineral]] because it is a key constituent of the respiratory enzyme complex [[cytochrome c oxidase]]. In [[molluscs]] and [[crustacea]]ns, copper is a constituent of the blood pigment [[hemocyanin]], replaced by the iron-complexed [[hemoglobin]] in fish and other [[vertebrate]]s. In humans, copper is found mainly in the liver, muscle, and bone.<ref>{{cite web |editor-last = Johnson, MD PhD |editor-first = Larry E. |title = Copper |work = Merck Manual Home Health Handbook |publisher = Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. |date = 2008 |url = http://www.merckmanuals.com/home/disorders_of_nutrition/minerals/copper.html |accessdate = 7 April 2013}}</ref> The adult body contains between 1.4 and 2.1 mg of copper per kilogram of body weight.<ref>{{cite web|url=http://www.copper.org/consumers/health/cu_health_uk.html|title=Copper in human health}}</ref> |
|||
==Characteristics== |
|||
===Physical=== |
|||
[[File:Cu-Scheibe.JPG|thumb|left|150px|A copper disc (99.95% pure) made by [[continuous casting]]; [[industrial etching|etched]] to reveal [[crystallite]]s.]] |
|||
[[File:Copper just above its melting point.jpg|left|150px|thumb|Copper just above its melting point keeps its pink luster color when enough light outshines the orange [[incandescence]] color.]] |
|||
Copper, [[silver]], and [[gold]] are in [[group 11 element|group 11]] of the periodic table; these three metals have one s-orbital electron on top of a filled d-[[electron shell]] and are characterized by high [[ductility]], and electrical and thermal conductivity. The filled d-shells in these elements contribute little to interatomic interactions, which are dominated by the s-electrons through [[metallic bond]]s. Unlike metals with incomplete d-shells, metallic bonds in copper are lacking a [[covalent bond|covalent]] character and are relatively weak. This observation explains the low [[hardness]] and high ductility of [[monocrystalline|single crystals]] of copper.<ref name=b1>{{cite book|author1=George L. Trigg|author2=Edmund H. Immergut|title=Encyclopedia of applied physics|url=https://books.google.com/books?id=sVQ5RAAACAAJ|accessdate=2 May 2011|date=1 November 1992|publisher=VCH Publishers|isbn=978-3-527-28126-8|pages=267–272|volume=4: Combustion to Diamagnetism}}</ref> At the macroscopic scale, introduction of extended defects to the [[crystal lattice]], such as grain boundaries, hinders flow of the material under applied stress, thereby increasing its hardness. For this reason, copper is usually supplied in a fine-grained [[polycrystalline]] form, which has greater strength than monocrystalline forms.<ref>{{cite book|author = Smith, William F.|author2 = Hashemi, Javad|last-author-amp = yes |title = Foundations of Materials Science and Engineering|page = 223|publisher = McGraw-Hill Professional|date= 2003|isbn = 0-07-292194-3}}</ref> |
|||
The softness of copper partly explains its high electrical conductivity (59.6×10<sup>6</sup> [[Siemens (unit)|S]]/m) and high thermal conductivity, second highest (second only to silver) among pure metals at room temperature.<ref name=CRC>{{cite book|author = Hammond, C. R.|title = The Elements, in Handbook of Chemistry and Physics|edition = 81st|publisher =CRC press|isbn = 0-8493-0485-7|date = 2004}}</ref> This is because the resistivity to electron transport in metals at room temperature originates primarily from scattering of electrons on thermal vibrations of the lattice, which are relatively weak in a soft metal.<ref name=b1/> The maximum permissible current density of copper in open air is approximately 3.1×10<sup>6</sup> A/m<sup>2</sup> of cross-sectional area, above which it begins to heat excessively.<ref>{{cite book|author=Resistance Welding Manufacturing Alliance |title=Resistance Welding Manual|date=2003|publisher=Resistance Welding Manufacturing Alliance|isbn=0-9624382-0-0|edition=4th|pages=18–12}}</ref> |
|||
Copper is one of a few metallic elements with a natural color other than gray or silver.<ref>{{Cite book|last = Chambers|first = William|last2 = Chambers|first2 = Robert|title = Chambers's Information for the People|publisher = W. & R. Chambers|date = 1884|volume = L|page = 312|edition = 5th|url = https://books.google.com/?id=eGIMAAAAYAAJ|isbn = 0-665-46912-8}}</ref> Pure copper is orange-red and acquires a reddish [[tarnish]] when exposed to air. The characteristic color of copper results from the electronic transitions between the filled 3d and half-empty 4s atomic shells – the energy difference between these shells corresponds to orange light. |
|||
As with other metals, if copper is put in contact with another metal, [[galvanic corrosion]] will occur.<ref>{{cite web|title=Galvanic Corrosion|url=http://www.corrosion-doctors.org/Forms-galvanic/galvanic-corrosion.htm|work=Corrosion Doctors|accessdate=29 April 2011}}</ref> |
|||
===Chemical=== |
|||
[[File:Copper wire comparison.JPG|left|150px|thumb|Unoxidized copper wire (left) and oxidized copper wire (right).]] |
|||
[[File:Royal Observatory Edinburgh East Tower 2010 cropped.jpg|thumb|The East Tower of the [[Royal Observatory, Edinburgh]]. The contrast between the refurbished copper installed in 2010 and the green color of the original 1894 copper is clearly seen.]] |
|||
Copper does not react with water, but it does slowly react with atmospheric oxygen to form a layer of brown-black copper oxide which, unlike the [[rust]] that forms on iron in moist air, protects the underlying metal from further corrosion ([[passivation (chemistry)|passivation]]). A green layer of [[verdigris]] (copper carbonate) can often be seen on old copper structures, such as the roofing of many older buildings<ref name=":0">{{Cite book|url=https://books.google.com/books?id=3qL3vfUZHMYC|title=Cultural Heritage Conservation and Environmental Impact Assessment by Non-Destructive Testing and Micro-Analysis|last=Grieken|first=Rene van|last2=Janssens|first2=Koen|date=2005-09-27|publisher=CRC Press|isbn=978-0-203-97078-2|page=197|language=en}}</ref> and the [[Statue of Liberty]].<ref>{{cite web|title=Copper.org: Education: Statue of Liberty: Reclothing the First Lady of Metals – Repair Concerns|url=http://www.copper.org/education/liberty/liberty_reclothed1.html|work=Copper.org|accessdate=11 April 2011}}</ref> Copper [[tarnish]]es when exposed to some [[sulfur]] compounds, with which it reacts to form various [[copper sulfide]]s.<ref>{{cite journal|last1=Rickett|first1=B. I.|last2=Payer|first2=J. H.|title=Composition of Copper Tarnish Products Formed in Moist Air with Trace Levels of Pollutant Gas: Hydrogen Sulfide and Sulfur Dioxide/Hydrogen Sulfide|journal=Journal of the Electrochemical Society|date=1995|volume=142|issue=11|pages=3723–3728|doi=10.1149/1.2048404}}</ref> |
|||
===Isotopes=== |
|||
{{Main article|Isotopes of copper}} |
|||
There are 29 [[isotope]]s of copper. <sup>63</sup>Cu and <sup>65</sup>Cu are stable, with <sup>63</sup>Cu comprising approximately 69% of naturally occurring copper; both have a [[spin (physics)|spin]] of {{frac|3|2}}.<ref name="nubase">{{cite journal|title=Nubase2003 Evaluation of Nuclear and Decay Properties|journal=Nuclear Physics A|volume=729|pages=3–128|publisher=Atomic Mass Data Center|date=2003|doi=10.1016/j.nuclphysa.2003.11.001|author=Audi, G|bibcode=2003NuPhA.729....3A|last2=Bersillon|first2=O.|last3=Blachot|first3=J.|last4=Wapstra|first4=A. H.}}</ref> The other isotopes are [[radioactivity|radioactive]], with the most stable being <sup>67</sup>Cu with a [[half-life]] of 61.83 hours.<ref name="nubase"/> Seven [[nuclear isomer|metastable isotopes]] have been characterized; <sup>68m</sup>Cu is the longest-lived with a half-life of 3.8 minutes. Isotopes with a [[mass number]] above 64 decay by [[beta decay|β<sup>−</sup>]], whereas those with a mass number below 64 decay by [[positron emission|β<sup>+</sup>]]. [[Copper-64|<sup>64</sup>Cu]], which has a half-life of 12.7 hours, decays both ways.<ref>{{cite web |url=http://www.nndc.bnl.gov/chart/reCenter.jsp?z=29&n=35 |title=Interactive Chart of Nuclides |work=National Nuclear Data Center |accessdate=8 April 2011}}</ref> |
|||
<sup>62</sup>Cu and <sup>64</sup>Cu have significant applications. <sup>62</sup>Cu is used in <sup>62</sup>Cu-PTSM as a [[radioactive tracer]] for [[positron emission tomography]].<ref>{{Cite journal | last = Okazawad | first = Hidehiko | last2 = Yonekura | first2 = Yoshiharu | last3 = Fujibayashi | first3 = Yasuhisa | last4 = Nishizawa | first4 = Sadahiko | last5 = Magata | first5 = Yasuhiro | last6 = Ishizu | first6 = Koichi | last7 = Tanaka | first7 = Fumiko | last8 = Tsuchida |first8 = Tatsuro |last9 = Tamaki |first9 = Nagara |last10 = Konishi | first10 = Junji |date=1994 |title=Clinical Application and Quantitative Evaluation of Generator-Produced Copper-62-PTSM as a Brain Perfusion Tracer for PET |journal=Journal of Nuclear Medicine |volume=35 |issue=12 |pages=1910–1915 |url=http://jnm.snmjournals.org/cgi/reprint/35/12/1910.pdf|pmid=7989968 |format=PDF}}</ref> |
|||
===Occurrence=== |
|||
<!--[[File:Native Copper from the Keweenaw Peninsula Michigan.jpg|thumb|right|150px|Native copper from the Keweenaw Peninsula Michigan about 2.5 inches (6.4 cm) long]]--> |
|||
Copper is produced in massive stars<ref>{{cite journal|last1=Romano|first1=Donatella|last2=Matteucci|first2=Fransesca|title=Contrasting copper evolution in ω Centauri and the Milky Way|journal=[[Monthly Notices of the Royal Astronomical Society]]: Letters|date=2007|volume=378|issue=1|pages=L59–L63|doi=10.1111/j.1745-3933.2007.00320.x|bibcode=2007MNRAS.378L..59R|arxiv = astro-ph/0703760}}</ref> and is present in the Earth's crust in a proportion of about 50 parts per million (ppm).<ref name=emsley>{{cite book|author=Emsley, John|title=Nature's building blocks: an A-Z guide to the elements|url=https://books.google.com/books?id=j-Xu07p3cKwC&pg=PA123|accessdate=2 May 2011|date=11 August 2003|publisher=Oxford University Press|isbn=978-0-19-850340-8|pages=121–125}}</ref> It occurs as [[native copper]], in the copper sulfides [[chalcopyrite]] and [[chalcocite]], in the copper carbonates [[azurite]] and [[malachite]], and in the [[copper(I) oxide]] mineral [[cuprite]].<ref name=CRC/> The largest mass of elemental copper discovered weighed 420 tonnes and was found in 1857 on the [[Keweenaw Peninsula]] in [[Michigan]], US.<ref name=emsley/> Native copper is a [[polycrystal]], with the largest single crystal ever described measuring 4.4×3.2×3.2 cm.<ref>{{cite journal|url = http://www.minsocam.org/ammin/AM66/AM66_885.pdf|journal = American Mineralogist|volume = 66|page=885|date= 1981|title= The largest crystals|author = Rickwood, P. C.}}</ref> |
|||
==Production== |
|||
[[File:Chuquicamata-002.jpg|thumb|left|[[Chuquicamata]] in Chile is one of the world's largest [[open-pit mining|open pit]] copper [[mining|mines]].]] |
|||
[[File:Copper - world production trend.svg|thumb|World production trend]] |
|||
[[File:Copper Price History USD.png|thumb|Copper prices 2003–2011 in USD per tonne]] |
|||
{{see also|List of countries by copper production}} |
|||
Most copper is mined or [[copper extraction techniques|extracted]] as copper sulfides from large [[open pit mine]]s in [[porphyry copper]] deposits that contain 0.4 to 1.0% copper. Sites include [[Chuquicamata]] in Chile, [[Bingham Canyon Mine]] in Utah, United States and [[El Chino Mine]] in New Mexico, United States. According to the [[British Geological Survey]] in 2005, Chile was the top producer of copper with at least one-third world share followed by the United States, Indonesia and Peru.<ref name=CRC/> Copper can also be recovered through the [[in-situ leach]] process. Several sites in the state of Arizona are considered prime candidates for this method.<ref>{{cite web|last=Randazzo |first=Ryan |url=http://www.azcentral.com/arizonarepublic/business/articles/2011/06/19/20110619copper-new-method-fight.html |title=A new method to harvest copper |publisher=Azcentral.com |date=19 June 2011 |accessdate=25 April 2014}}</ref> The amount of copper in use is increasing and the quantity available is barely sufficient to allow all countries to reach developed world levels of usage.<ref>{{cite journal|url=http://www.pnas.org/content/103/5/1209.full|title=Metal stocks and sustainability|journal=Proceedings of the National Academy of Sciences |date=2006|volume=103|issue=5|pages=1209–1214|first1=R. B.|last1=Gordon|first2=M.|last2=Bertram|first3=T. E.|last3=Graedel|doi=10.1073/pnas.0509498103|pmc=1360560|pmid=16432205|bibcode = 2006PNAS..103.1209G}}</ref> |
|||
===Reserves=== |
|||
{{see also|Peak copper#Reserves}} |
|||
Copper has been in use at least 10,000 years, but more than 95% of all copper ever mined and [[smelting|smelted]] has been extracted since 1900,<ref name=Leonard2006/> and more than half was extracted the last 24 years. As with many natural resources, the total amount of copper on Earth is vast, with around 10<sup>14</sup> tons in the top kilometer of Earth's crust, which is about 5 million years' worth at the current rate of extraction. However, only a tiny fraction of these reserves is economically viable with present-day prices and technologies. Estimates of copper reserves available for mining vary from 25 years to 60 years, depending on core assumptions such as the growth rate.<ref>{{cite book|author=Brown, Lester|title=Plan B 2.0: Rescuing a Planet Under Stress and a Civilization in Trouble|publisher=New York: W.W. Norton|date=2006|page=109|isbn=0-393-32831-7}}</ref> Recycling is a major source of copper in the modern world.<ref name=Leonard2006>{{cite web|url=http://www.salon.com/tech/htww/2006/03/02/peak_copper/index.html |title=Peak copper? |publisher=Salon – How the World Works |author=Leonard, Andrew |date=2 March 2006 |accessdate=23 March 2008 |deadurl=yes |archiveurl=https://web.archive.org/web/20080307042349/http://www.salon.com/tech/htww/2006/03/02/peak_copper/index.html |archivedate=7 March 2008 |df=dmy }}</ref> Because of these and other factors, the future of copper production and supply is the subject of much debate, including the concept of [[peak copper]], analogous to [[peak oil]]. |
|||
The price of copper has historically been unstable,<ref>{{cite journal|last=Schmitz|first=Christopher|title=The Rise of Big Business in the World, Copper Industry 1870–1930|journal=Economic History Review|date=1986|volume=39|series=2|issue=3|pages=392–410|jstor=2596347|doi=10.1111/j.1468-0289.1986.tb00411.x}}</ref> and it sextupled from the 60-year low of US$0.60/lb (US$1.32/kg) in June 1999 to US$3.75 per pound (US$8.27/kg) in May 2006. It dropped to US$2.40/lb (US$5.29/kg) in February 2007, then rebounded to US$3.50/lb (US$7.71/kg) in April 2007.<ref>{{cite web|url = http://metalspotprice.com/copper-trends/|title =Copper Trends: Live Metal Spot Prices}}</ref>{{Better source|reason=This page is useless for verification as it is updated hourly. Archives exist, but they do not go back far enough and in any case the graphs are not shown with the precision quoted|date=April 2014}} In February 2009, weakening global demand and a steep fall in commodity prices since the previous year's highs left copper prices at US$1.51/lb (US$3.32/kg).<ref>{{cite news|url = http://www.forbes.com/2009/02/04/copper-frontera-southern-markets-equity-0205_china_51.html|title = A Bottom In Sight For Copper|author = Ackerman, R. |date = 2 April 2009|publisher = Forbes}}</ref> |
|||
===Methods=== |
|||
{{main article|Copper extraction techniques}} |
|||
[[File:Copper Flash Smelting Process (EN).svg|left|thumb|Scheme of flash smelting process]] |
|||
The concentration of copper in ores averages only 0.6%, and most commercial ores are sulfides, especially chalcopyrite (CuFeS<sub>2</sub>) and to a lesser extent chalcocite (Cu<sub>2</sub>S).<ref name=G&E/> These minerals are concentrated from [[comminution|crushed]] ores to the level of 10–15% copper by [[froth flotation]] or [[bioleaching]].<ref>{{cite journal|last=Watling |first=H. R. |title=The bioleaching of sulphide minerals with emphasis on copper sulphides — A review |journal=Hydrometallurgy |date=2006 |volume=84 |issue=1, 2 |pages=81–108 |url=http://infolib.hua.edu.vn/Fulltext/ChuyenDe/ChuyenDe07/CDe53/59.pdf |format=PDF |doi=10.1016/j.hydromet.2006.05.001 |deadurl=yes |archiveurl=https://web.archive.org/web/20110818131019/http://infolib.hua.edu.vn/Fulltext/ChuyenDe/ChuyenDe07/CDe53/59.pdf |archivedate=18 August 2011 |df=dmy }}</ref> Heating this material with [[silica]] in [[flash smelting]] removes much of the iron as [[slag]]. The process exploits the greater ease of converting iron sulfides into oxides, which in turn react with the silica to form the [[silicate]] slag that floats on top of the heated mass. The resulting ''copper matte,'' consisting of Cu<sub>2</sub>S, is [[roasting (metallurgy)|roasted]] to convert all sulfides into oxides:<ref name=G&E>{{Greenwood&Earnshaw2nd}}</ref> |
|||
:2 Cu<sub>2</sub>S + 3 O<sub>2</sub> → 2 Cu<sub>2</sub>O + 2 SO<sub>2</sub> |
|||
The cuprous oxide is converted to ''blister'' copper upon heating: |
|||
:2 Cu<sub>2</sub>O → 4 Cu + O<sub>2</sub> |
|||
The Sudbury [[Matte (metallurgy)|matte]] process converted only half the sulfide to oxide and then used this oxide to remove the rest of the sulfur as oxide. It was then electrolytically refined and the anode mud exploited for the [[platinum]] and gold it contained. This step exploits the relatively easy reduction of copper oxides to copper metal. [[Natural gas]] is blown across the blister to remove most of the remaining oxygen and [[electrorefining]] is performed on the resulting material to produce pure copper:<ref>{{cite book|last=Samans|first=Carl|title=Engineering metals and their alloys|date=1949|publisher=Macmillan|location=New York|oclc=716492542}}</ref> |
|||
:Cu<sup>2+</sup> + 2 e<sup>−</sup> → Cu |
|||
{{Plain image|File:Ural Mining and Metallurgical Company Copper Map.svg|<big>'''Flowchart of copper refining''' (Anode casting plant of Uralelektromed)</big> {{columns-list|2| |
|||
# ''[[Copper extraction techniques#Converting|Blister copper]]'' |
|||
# ''[[Smelting]]'' |
|||
# ''[[Reverberatory furnace]]'' |
|||
# ''[[Slag]] removal'' |
|||
# ''Copper casting of [[anode]]s'' |
|||
# ''Casting wheel'' |
|||
# ''Anodes removal machine'' |
|||
# ''Anodes take-off'' |
|||
# ''[[Minecart|Rail cars]]'' |
|||
# ''Transportation to the tank house''}}|600px|center|top|triangle|#ccc}} |
|||
===Recycling=== |
|||
Like [[aluminium]],<ref>{{Cite book|url=https://books.google.com/books?id=Je4GBwAAQBAJ&pg=PT89&dq=Like+aluminium,+copper+recyclable+without+any+loss+of+quality&hl=en&sa=X&ved=0ahUKEwid0Zq524TVAhVU22MKHdW9ACYQ6AEITTAH#v=onepage&q=Like%20aluminium,%20copper%20recyclable%20without%20any%20loss%20of%20quality&f=false|title=Pen to Paper: Making Fun of Life|last=Burton|first=Julie McCulloch|date=2015-02-20|publisher=iUniverse|isbn=9781491753941|language=en}}</ref> copper is recyclable without any loss of quality, both from raw state and from manufactured products.<ref>{{Cite book|url=https://books.google.com/books?id=5_QLBwAAQBAJ&pg=PA249&dq=copper+recyclable+without+any+loss+of+quality&hl=en&sa=X&ved=0ahUKEwiEtPPq3ITVAhUM8WMKHfdrCCMQ6AEILjAC#v=onepage&q=copper%20recyclable%20without%20any%20loss%20of%20quality&f=false|title=The Role of Ecological Chemistry in Pollution Research and Sustainable Development|last=Bahadir|first=Ali Mufit|last2=Duca|first2=Gheorghe|date=2009-08-03|publisher=Springer|isbn=9789048129034|language=en}}</ref> In volume, copper is the third most recycled metal after iron and aluminium.<ref>{{Cite book|url=https://books.google.com/books?id=BnN3DAAAQBAJ&pg=PT281&dq=%C2%A0copper+third+most+recycled+metal+after+iron+and+aluminium&hl=en&sa=X&ved=0ahUKEwjGuq2S3YTVAhUP92MKHc5WBjEQ6AEILDAC#v=onepage&q=%C2%A0copper%20third%20most%20recycled%20metal%20after%20iron%20and%20aluminium&f=false|title=The Periodic Table in Minutes|last=Green|first=Dan|date=2016-09-06|publisher=Quercus|isbn=9781681443294|language=en}}</ref> An estimated 80% of all copper ever mined is still in use today.<ref>{{cite web|url=http://www.copperinfo.com/environment/recycling.html|title=International Copper Association}}</ref> According to the [[International Resource Panel]]'s [[Metal Stocks in Society report]], the global per capita stock of copper in use in society is 35–55 kg. Much of this is in more-developed countries (140–300 kg per capita) rather than less-developed countries (30–40 kg per capita). |
|||
The process of recycling copper is roughly the same as is used to extract copper but requires fewer steps. High-purity scrap copper is melted in a [[furnace#Metallurgical furnace|furnace]] and then [[redox|reduced]] and cast into [[Billet (semi-finished product)|billets]] and [[ingot]]s; lower-purity scrap is refined by [[electroplating]] in a bath of [[sulfuric acid]].<ref>[http://www.copper.org/publications/newsletters/innovations/1998/06/recycle_overview.html "Overview of Recycled Copper" ''Copper.org'']. Copper.org (25 August 2010). Retrieved on 8 November 2011.</ref> |
|||
==Alloys== |
|||
{{See also|List of copper alloys}} |
|||
Numerous copper [[alloy]]s have been formulated, many with important uses. [[Brass]] is an alloy of copper and [[zinc]]. [[Bronze]] usually refers to copper-[[tin]] alloys, but can refer to any alloy of copper such as [[aluminium bronze]]. Copper is one of the most important constituents of silver and [[carat (purity)|carat]] gold and carat solders used in the jewelry industry, modifying the color, hardness and melting point of the resulting alloys.<ref name=goldalloys>{{cite web|url=http://www.utilisegold.com/jewellery_technology/colours/colour_alloys/ |accessdate=6 June 2009 |title=Gold Jewellery Alloys |publisher=World Gold Council |deadurl=yes |archiveurl=https://web.archive.org/web/20090414151414/http://www.utilisegold.com/jewellery_technology/colours/colour_alloys |archivedate=14 April 2009 |df=dmy }}</ref> Some lead-free [[solder#Solder alloys|solders]] consist of tin alloyed with a small proportion of copper and other metals.<ref>[http://www.balverzinn.com/downloads/Solder_Sn97Cu3.pdf Balver Zinn Solder Sn97Cu3] {{webarchive |url=https://web.archive.org/web/20110707210148/http://www.balverzinn.com/downloads/Solder_Sn97Cu3.pdf |date=7 July 2011 }}. (PDF) . balverzinn.com. Retrieved on 8 November 2011.</ref> |
|||
The alloy of copper and [[nickel]], called [[cupronickel]], is used in low-denomination coins, often for the outer cladding. The US 5-cent coin (currently called a ''nickel'') consists of 75% copper and 25% nickel in homogeneous composition. The alloy of 90% copper and 10% nickel, remarkable for its resistance to corrosion, is used for various objects exposed to seawater, though it is vulnerable to the sulfides sometimes found in polluted harbors and estuaries.<ref>{{Cite book|url=https://books.google.com/books?id=8C7pXhnqje4C|title=Corrosion Tests and Standards|publisher=ASTM International|page=368|language=en}}</ref> Alloys of copper with aluminium (about 7%) have a golden color and are used in decorations.<ref name=emsley/> [[Shakudō]] is a Japanese decorative alloy of copper containing a low percentage of gold, typically 4–10%, that can be [[patina]]ted to a dark blue or black color.<ref name="Shakudō">{{cite journal|last=Oguchi|first=Hachiro|date=1983|title=Japanese Shakudō: its history, properties and production from gold-containing alloys|journal=Gold Bulletin|volume=16|issue=4|pages=125–132|publisher=World Gold Council|doi=10.1007/BF03214636}}<!--|accessdate=4 June 2016 --></ref> |
|||
==Compounds== |
|||
[[File:CopperIoxide.jpg|thumb|A sample of [[copper(I) oxide]].]] |
|||
{{See also|Category:Copper compounds}} |
|||
Copper forms a rich variety of compounds, usually with [[oxidation state]]s +1 and +2, which are often called ''cuprous'' and ''cupric'', respectively.<ref name="Holleman"/> |
|||
===Binary compounds=== |
|||
As with other elements, the simplest compounds of copper are binary compounds, i.e. those containing only two elements, the principal examples being oxides, sulfides, and [[halide]]s. Both [[copper(I) oxide|cuprous]] and [[copper(II) oxide|cupric oxides]] are known. Among the numerous [[copper sulfide]]s, important examples include [[copper(I) sulfide]] and [[copper monosulfide|copper(II) sulfide]]. |
|||
Cuprous halides (with [[copper(I) chloride|chlorine]], [[copper(I) bromide|bromine]], and [[copper(I) iodide|iodine]]) are known, as are cupric halides with [[copper(II) fluoride|fluorine]], [[copper(II) chloride|chlorine]], and [[copper(II) bromide|bromine]]. Attempts to prepare copper(II) iodide yield only cuprous iodide and iodine.<ref name="Holleman">{{cite book |last1=Holleman |first1=A. F. |last2=Wiberg |first2=N. |title=Inorganic Chemistry |date=2001 |publisher=Academic Press |location=San Diego |isbn=978-0-12-352651-9}}</ref> |
|||
:2 Cu<sup>2+</sup> + 4 I<sup>−</sup> → 2 CuI + I<sub>2</sub> |
|||
===Coordination chemistry=== |
|||
[[File:Tetramminkupfer(II)-sulfat-Monohydrat Kristalle.png|thumb|left|Copper(II) gives a deep blue coloration in the presence of ammonia ligands. The one used here is [[Tetraamminecopper(II) sulfate|tetramminecopper(II) sulfate]].]] |
|||
Copper forms [[coordination complex]]es with [[ligand]]s. In aqueous solution, copper(II) exists as [Cu(H<sub>2</sub>O)<sub>6</sub>]<sup>2+</sup>. This complex exhibits the fastest water exchange rate (speed of water ligands attaching and detaching) for any transition [[metal aquo complex]]. Adding aqueous [[sodium hydroxide]] causes the precipitation of light blue solid [[copper(II) hydroxide]]. A simplified equation is: [[File:Cu-pourbaix-diagram.svg|thumbnail|Pourbaix diagram for copper in uncomplexed media (anions not other than OH- considered). Ion concentration 0.001 m (mol/kg water). Temperature 25 °C.]] |
|||
:Cu<sup>2+</sup> + 2 OH<sup>−</sup> → Cu(OH)<sub>2</sub> |
|||
[[Ammonia solution|Aqueous ammonia]] results in the same precipitate. Upon adding excess ammonia, the precipitate dissolves, forming [[Schweizer's reagent|tetraamminecopper(II)]]: |
|||
:Cu(H<sub>2</sub>O)<sub>4</sub>(OH)<sub>2</sub> + 4 NH<sub>3</sub> → [Cu(H<sub>2</sub>O)<sub>2</sub>(NH<sub>3</sub>)<sub>4</sub>]<sup>2+</sup> + 2 H<sub>2</sub>O + 2 OH<sup>−</sup> |
|||
Many other [[oxyanion]]s form complexes; these include [[copper(II) acetate]], [[copper(II) nitrate]], and [[copper(II) carbonate]]. [[Copper(II) sulfate]] forms a blue crystalline penta[[hydrate]], the most familiar copper compound in the laboratory. It is used in a [[fungicide]] called the [[Bordeaux mixture]].<ref name="Boux">{{cite book|url = https://books.google.com/books?id=cItuoO9zSjkC&pg=PA623|page = 623|chapter = Nonsystematic (Contact) Fungicides|title = Ullmann's Agrochemicals|isbn = 978-3-527-31604-5|author1 = Wiley-Vch|date = 2 April 2007}}</ref> |
|||
[[File:Tetraamminediaquacopper(II)-3D-balls.png|thumb|right|200px|[[Ball-and-stick model]] of the complex [Cu(NH<sub>3</sub>)<sub>4</sub>(H<sub>2</sub>O)<sub>2</sub>]<sup>2+</sup>, illustrating the [[octahedral coordination geometry]] common for copper(II).]] |
|||
[[Polyol]]s, compounds containing more than one alcohol [[functional group]], generally interact with cupric salts. For example, copper salts are used to test for [[reducing sugars]]. Specifically, using [[Benedict's reagent]] and [[Fehling's solution]] the presence of the sugar is signaled by a color change from blue Cu(II) to reddish copper(I) oxide.<ref>Ralph L. Shriner, Christine K. F. Hermann, Terence C. Morrill, David Y. Curtin, Reynold C. Fuson "The Systematic Identification of Organic Compounds" 8th edition, J. Wiley, Hoboken. {{ISBN|0-471-21503-1}}</ref> Schweizer's reagent and related complexes with [[ethylenediamine]] and other [[amine]]s dissolve [[cellulose]].<ref>{{cite journal | last1 = Saalwächter | first1 = Kay | last2 = Burchard | first2 = Walther | last3 = Klüfers | first3 = Peter | last4 = Kettenbach | first4 = G. | last5 = Mayer | first5 = Peter | last6 = Klemm | first6 = Dieter | last7 = Dugarmaa | first7 = Saran | year = 2000 | title = Cellulose Solutions in Water Containing Metal Complexes | url = | journal = Macromolecules | volume = 33 | issue = | pages = 4094–4107 | doi = 10.1021/ma991893m }}</ref> [[Amino acid]]s form very stable [[chelate complex]]es with copper(II). Many wet-chemical tests for copper ions exist, one involving [[potassium ferrocyanide]], which gives a brown precipitate with copper(II) salts. |
|||
===Organocopper chemistry=== |
|||
{{Main article|Organocopper compound}} |
|||
Compounds that contain a carbon-copper bond are known as organocopper compounds. They are very reactive towards oxygen to form copper(I) oxide and have [[Reactions of organocopper reagents|many uses in chemistry]]. They are synthesized by treating copper(I) compounds with [[Grignard reaction|Grignard reagents]], [[terminal alkyne]]s or [[organolithium compound|organolithium reagents]];<ref>"Modern Organocopper Chemistry" Norbert Krause, Ed., Wiley-VCH, Weinheim, 2002. {{ISBN|978-3-527-29773-3}}.</ref> in particular, the last reaction described produces a [[Gilman reagent]]. These can undergo [[substitution reaction|substitution]] with [[alkyl halides]] to form [[coupling reaction|coupling products]]; as such, they are important in the field of [[organic synthesis]]. [[Copper(I) acetylide]] is highly shock-sensitive but is an intermediate in reactions such as the [[Cadiot-Chodkiewicz coupling]]<ref>{{cite journal|last1=Berná|first1=José|last2=Goldup|first2=Stephen|last3=Lee|first3=Ai-Lan|last4=Leigh|first4=David|last5=Symes|first5=Mark|last6=Teobaldi|first6=Gilberto|last7=Zerbetto|first7=Fransesco|title=Cadiot–Chodkiewicz Active Template Synthesis of Rotaxanes and Switchable Molecular Shuttles with Weak Intercomponent Interactions|journal=Angewandte Chemie|date=26 May 2008|volume=120|issue=23|pages=4464–4468|doi=10.1002/ange.200800891}}</ref> and the [[Sonogashira coupling]].<ref>{{cite journal|title = The Sonogashira Reaction: A Booming Methodology in Synthetic Organic Chemistry|author = Rafael Chinchilla|author2 = Carmen Nájera|last-author-amp = yes|journal = [[Chemical Reviews]]|date = 2007|volume = 107|issue = 3|pages = 874–922|doi = 10.1021/cr050992x|pmid = 17305399}}</ref> [[Nucleophilic conjugate addition|Conjugate addition]] to [[enone]]s<ref>{{cite journal|date=1986 |title=An Addition of an Ethylcopper Complex to 1-Octyne: (E)-5-Ethyl-1,4-Undecadiene |journal=[[Organic Syntheses]] |volume=64 |page=1 |url=http://www.orgsyn.org/orgsyn/pdfs/CV7P0236.pdf |format=PDF |doi=10.15227/orgsyn.064.0001 |deadurl=yes |archiveurl=https://web.archive.org/web/20120619005340/http://www.orgsyn.org/orgsyn/pdfs/CV7P0236.pdf |archivedate=19 June 2012 |df=dmy }}</ref> and [[carbometalation|carbocupration]] of alkynes<ref>{{cite journal |last1=Kharasch |first1=M. S. |last2=Tawney |first2=P. O. |date=1941|title=Factors Determining the Course and Mechanisms of Grignard Reactions. II. The Effect of Metallic Compounds on the Reaction between Isophorone and Methylmagnesium Bromide |journal=Journal of the American Chemical Society |volume=63 |issue=9 |pages=2308–2316 |doi=10.1021/ja01854a005}}</ref> can also be achieved with organocopper compounds. Copper(I) forms a variety of weak complexes with [[alkene]]s and [[carbon monoxide]], especially in the presence of amine ligands.<ref>{{cite journal|last= Imai |first= Sadako |last2= Fujisawa |first2= Kiyoshi |last3= Kobayashi |first3= Takako |last4= Shirasawa |first4= Nobuhiko |last5= Fujii |first5= Hiroshi |last6= Yoshimura |first6= Tetsuhiko |last7= Kitajima |first7= Nobumasa |last8= Moro-oka |first8= Yoshihiko |title= <sup>63</sup>Cu NMR Study of Copper(I) Carbonyl Complexes with Various Hydrotris(pyrazolyl)borates: Correlation between 63Cu Chemical Shifts and CO Stretching Vibrations|journal= Inorganic Chemistry |date= 1998| volume =37|pages=3066–3070|doi=10.1021/ic970138r|issue=12}}</ref> |
|||
===Copper(III) and copper(IV)=== |
|||
Copper(III) is most often found in oxides. A simple example is potassium [[cuprate]], KCuO<sub>2</sub>, a blue-black solid.<ref>{{cite book|chapter=Potassium Cuprate (III)|title=Handbook of Preparative Inorganic Chemistry|edition=2nd|editor=G. Brauer|publisher=Academic Press|year=1963|location=NY|volume=1|page=1015}}</ref> The most extensively studied copper(III) compounds are the [[cuprate superconductor]]s. [[Yttrium barium copper oxide]] (YBa<sub>2</sub>Cu<sub>3</sub>O<sub>7</sub>) consists of both Cu(II) and Cu(III) centres. Like oxide, [[fluoride]] is a highly [[base (chemistry)|basic]] [[anion]]<ref>{{cite journal|author1=Schwesinger, Reinhard |author2=Link, Reinhard |author3=Wenzl, Peter |author4=Kossek, Sebastian |title=Anhydrous phosphazenium fluorides as sources for extremely reactive fluoride ions in solution|doi=10.1002/chem.200500838|year=2006|journal=Chemistry: A European Journal|volume=12|issue=2|pages=438–45 |pmid=16196062}}</ref> and is known to stabilize metal ions in high oxidation states. Both copper(III) and even copper(IV) fluorides are known, [[Potassium hexafluorocuprate(III)|K<sub>3</sub>CuF<sub>6</sub>]] and [[Caesium hexafluorocuprate(IV)|Cs<sub>2</sub>CuF<sub>6</sub>]], respectively.<ref name=Holleman/> |
|||
Some copper proteins form [[oxo complex]]es, which also feature copper(III).<ref>{{cite journal |last1=Lewis |first1=E. A. |last2=Tolman |first2=W. B. |date=2004 |title=Reactivity of Dioxygen-Copper Systems |journal=Chemical Reviews |volume=104 |pages=1047–1076 |doi=10.1021/cr020633r |issue=2 |pmid=14871149}}</ref> With [[tetrapeptide]]s, purple-colored copper(III) complexes are stabilized by the deprotonated [[amide]] ligands.<ref>{{cite journal |last1=McDonald |first1=M. R. |last2=Fredericks |first2=F. C. |last3=Margerum |first3=D. W. |date=1997 |title=Characterization of Copper(III)-Tetrapeptide Complexes with Histidine as the Third Residue |journal=Inorganic Chemistry |volume=36 |pages=3119–3124|doi=10.1021/ic9608713|pmid=11669966 |issue=14}}</ref> |
|||
Complexes of copper(III) are also found as intermediates in reactions of organocopper compounds.<ref>{{Greenwood&Earnshaw2nd|page=1187}}</ref> |
|||
==History== |
|||
===Copper Age=== |
|||
{{Main article|Copper Age}} |
|||
[[File:Minoan copper ingot from Zakros, Crete.jpg|left|thumb|A corroded copper [[ingot]] from [[Zakros]], [[Crete]], shaped in the form of an animal skin typical in that era.]] |
|||
[[File:ReconstructedOetziAxe.jpg|thumb|Many tools during the [[Chalcolithic]] Era included copper, such as the blade of this replica of [[Ötzi]]'s axe]] |
|||
[[File:Chrysocolla Timna 070613.jpg|left|thumb|Copper ore ([[chrysocolla]]) in [[Cambrian]] sandstone from [[Chalcolithic]] mines in the [[Timna Valley]], southern [[Israel]].]] |
|||
Copper occurs naturally as [[native copper|native metallic copper]] and was known to some of the oldest civilizations on record. The history of copper use dates to 9000 BC in the Middle East;<ref name=discovery>{{cite web|url=http://www.csa.com/discoveryguides/copper/overview.php|title=CSA – Discovery Guides, A Brief History of Copper|publisher=Csa.com|accessdate=12 September 2008}}</ref> a copper pendant was found in northern Iraq that dates to 8700 BC.<ref>{{cite book|page = 56|title = Jewelrymaking through History: an Encyclopedia|publisher= Greenwood Publishing Group|date = 2007|isbn = 0-313-33507-9|author = Rayner W. Hesse}}No primary source is given in that book.</ref> Evidence suggests that gold and [[meteoric iron]] (but not iron smelting) were the only metals used by humans before copper.<ref name=vander>{{cite web|url=http://elements.vanderkrogt.net/element.php?sym=Cu|title=Copper|publisher=Elements.vanderkrogt.net|accessdate=12 September 2008}}</ref> The history of copper metallurgy is thought to follow this sequence: First, [[Work hardening|cold working]] of native copper, then [[Annealing (metallurgy)|annealing]], [[smelting]], and, finally, [[lost-wax casting]]. In southeastern [[Anatolia]], all four of these techniques appear more or less simultaneously at the beginning of the [[Neolithic]] c. 7500 BC.<ref name="Renfrew1990">{{cite book|last=Renfrew|first=Colin|authorlink=Colin Renfrew, Baron Renfrew of Kaimsthorn|title=Before civilization: the radiocarbon revolution and prehistoric Europe|url=https://books.google.com/books?id=jJhHPgAACAAJ|accessdate=21 December 2011|date=1990|publisher=Penguin|isbn=978-0-14-013642-5}}</ref> |
|||
Copper smelting was independently invented in different places. It was probably discovered in China before 2800 BC, in Central America around 600 AD, and in West Africa about the 9th or 10th century AD.<ref>{{cite news|author = Cowen, R. |url = http://www.geology.ucdavis.edu/~cowen/~GEL115/115CH3.html|title = Essays on Geology, History, and People: Chapter 3: Fire and Metals|accessdate =7 July 2009}}</ref> [[Investment casting]] was invented in 4500–4000 BC in Southeast Asia<ref name="discovery" /> and [[carbon dating]] has established mining at [[Alderley Edge]] in [[Cheshire]], UK, at 2280 to 1890 BC.<ref>{{cite book|author = Timberlake, S.|author2 = Prag A.J.N.W.|last-author-amp = yes|date = 2005|title = The Archaeology of Alderley Edge: Survey, excavation and experiment in an ancient mining landscape|location = Oxford|publisher = John and Erica Hedges Ltd.|page = 396}}</ref> [[Ötzi the Iceman]], a male dated from 3300–3200 BC, was found with an axe with a copper head 99.7% pure; high levels of [[arsenic]] in his hair suggest an involvement in copper smelting.<ref name="CSA">{{cite web|title=CSA – Discovery Guides, A Brief History of Copper|url=http://www.csa.com/discoveryguides/copper/overview.php|work=CSA Discovery Guides|accessdate=29 April 2011}}</ref> Experience with copper has assisted the development of other metals; in particular, copper smelting led to the discovery of [[bloomery|iron smelting]].<ref name="CSA" /> Production in the [[Old Copper Complex]] in Michigan and Wisconsin is dated between 6000 and 3000 BC.<ref name="occ">Pleger, Thomas C. "A Brief Introduction to the Old Copper Complex of the Western Great Lakes: 4000–1000 BC", ''[https://books.google.com/books?id=6NUQNQAACAAJ Proceedings of the Twenty-seventh Annual Meeting of the Forest History Association of Wisconsin]'', Oconto, Wisconsin, 5 October 2002, pp. 10–18.</ref><ref>Emerson, Thomas E. and McElrath, Dale L. ''[https://books.google.com/books?id=awsA08oYoskC&pg=PA709 Archaic Societies: Diversity and Complexity Across the Midcontinent]'', SUNY Press, 2009 {{ISBN|1-4384-2701-8}}.</ref> Natural bronze, a type of copper made from ores rich in silicon, arsenic, and (rarely) tin, came into general use in the Balkans around 5500 BC.{{citation needed|date=April 2014}} |
|||
===Bronze Age=== |
|||
{{Main article|Bronze Age}} |
|||
Alloying copper with tin to make bronze was first practiced about 4000 years after the discovery of copper smelting, and about 2000 years after "natural bronze" had come into general use.{{citation needed|date=September 2015}} Bronze artifacts from the [[Vinča culture]] date to 4500 BC.<ref name=antiquity1312>{{cite web | url = http://antiquity.ac.uk/ant/087/ant0871030.htm | title = Tainted ores and the rise of tin bronzes in Eurasia, c. 6500 years ago | first = Miljana | last = Radivojević | first2 = Thilo | last2 = Rehren | publisher = Antiquity Publications Ltd | date = December 2013}}</ref> [[Sumer]]ian and [[Ancient Egypt|Egyptian]] artifacts of copper and bronze alloys date to 3000 BC.<ref name=hist>{{cite book|pages = 13, 48–66|title = Encyclopaedia of the History of Technology|author = McNeil, Ian |publisher = Routledge|date = 2002|location = London ; New York|isbn = 0-203-19211-7}}</ref> The [[Bronze Age]] began in Southeastern Europe around 3700–3300 BC, in Northwestern Europe about 2500 BC. It ended with the beginning of the Iron Age, 2000–1000 BC in the Near East, and 600 BC in Northern Europe. The transition between the [[Neolithic]] period and the Bronze Age was formerly termed the [[Chalcolithic]] period (copper-stone), when copper tools were used with stone tools. The term has gradually fallen out of favor because in some parts of the world, the Chalcolithic and Neolithic are coterminous at both ends. Brass, an alloy of copper and zinc, is of much more recent origin. It was known to the Greeks, but became a significant supplement to bronze during the Roman Empire.<ref name=hist/> |
|||
===Antiquity and Middle Ages=== |
|||
[[File:Venus symbol.svg|thumb|left|100px|In [[alchemy]] the symbol for copper was also the symbol for the goddess and planet [[Venus]].]] |
|||
[[File:TimnaChalcolithicMine.JPG|thumb|Chalcolithic copper mine in [[Timna Valley]], [[Negev Desert]], Israel.]] |
|||
In Greece, copper was known by the name ''chalkos'' (χαλκός). It was an important resource for the Romans, Greeks and other ancient peoples. In Roman times, it was known as ''aes Cyprium'', ''aes'' being the generic Latin term for copper alloys and ''Cyprium'' from [[Cyprus]], where much copper was mined. The phrase was simplified to ''cuprum'', hence the English ''copper''. [[Aphrodite]] ([[Venus (goddess)|Venus]] in Rome) represented copper in mythology and alchemy because of its lustrous beauty and its ancient use in producing mirrors; Cyprus was sacred to the goddess. The seven heavenly bodies known to the ancients were associated with the seven metals known in antiquity, and Venus was assigned to copper.<ref>{{cite journal|title = The Nomenclature of Copper and its Alloys|author = Rickard, T. A. |journal = Journal of the Royal Anthropological Institute|volume = 62|date = 1932|page=281|jstor = 2843960|doi = 10.2307/2843960|publisher = Royal Anthropological Institute}}</ref> |
|||
Copper was first used in ancient Britain in about the 3rd or 2nd Century BC. In North America, copper mining began with marginal workings by Native Americans. Native copper is known to have been extracted from sites on [[Isle Royale]] with primitive stone tools between 800 and 1600.<ref>{{cite journal|title = The State of Our Knowledge About Ancient Copper Mining in Michigan|journal = The Michigan Archaeologist|volume = 41|page = 119|author = Martin, Susan R. |date = 1995|url = http://www.ramtops.co.uk/copper.html|issue =2–3}}</ref> Copper metallurgy was flourishing in South America, particularly in Peru around 1000 AD. Copper burial ornamentals from the 15th century have been uncovered, but the metal's commercial production did not start until the early 20th century. |
|||
The cultural role of copper has been important, particularly in currency. Romans in the 6th through 3rd centuries BC used copper lumps as money. At first, the copper itself was valued, but gradually the shape and look of the copper became more important. [[Julius Caesar]] had his own coins made from brass, while [[Augustus|Octavianus Augustus Caesar]]'s coins were made from Cu-Pb-Sn alloys. With an estimated annual output of around 15,000 t, [[Roman metallurgy|Roman copper mining and smelting activities]] reached a scale unsurpassed until the time of the [[Industrial Revolution]]; the [[Roman province|provinces]] most intensely mined were those of [[Hispania]], [[Cyprus]] and in Central Europe.<ref>{{cite journal|doi = 10.1126/science.272.5259.246|title = History of Ancient Copper Smelting Pollution During Roman and Medieval Times Recorded in Greenland Ice|pages = 246–249 (247f.)|date = 1996|last1 = Hong|first1 = S.|last2 = Candelone|first2 = J.-P.|issue = 5259|last3 = Patterson|first3 = C. C.|last4 = Boutron|first4 = C. F.|journal = Science|volume = 272|bibcode = 1996Sci...272..246H}}</ref><ref>{{cite journal|last = de Callataÿ|first = François|date = 2005|title = The Graeco-Roman Economy in the Super Long-Run: Lead, Copper, and Shipwrecks|journal = Journal of Roman Archaeology|volume = 18|pages = 361–372 (366–369)}}</ref> |
|||
The gates of the [[Temple of Jerusalem]] used [[Corinthian bronze]] treated with [[depletion gilding]].{{Clarify|reason=Bronze is not a gold alloy, but depletion gilding can be done only on gold alloy.|date=June 2016}}{{Citation needed|date=June 2016}} The process was most prevalent in [[Alexandria]], where alchemy is thought to have begun.<ref>{{cite journal|url=http://www.goldbulletin.org/downloads/JACOB_2_33.PDF |title=Corinthian Bronze and the Gold of the Alchemists |author=Savenije, Tom J. |author2=Warman, John M. |author3=Barentsen, Helma M. |author4=van Dijk, Marinus |author5=Zuilhof, Han |author6=Sudhölter, Ernst J. R. |journal=Macromolecules |issue=2 |volume=33 |date=2000 |pages=60–66 |doi=10.1021/ma9904870 |bibcode=2000MaMol..33...60S |deadurl=yes |archiveurl=https://web.archive.org/web/20070929003743/http://www.goldbulletin.org/downloads/JACOB_2_33.PDF |archivedate=29 September 2007 }}</ref> In ancient [[India]], copper was used in the [[holistic]] medical science [[Ayurveda]] for [[surgical]] instruments and other medical equipment. [[Ancient Egypt]]ians ([[Old Kingdom|~2400 BC]]) used copper for sterilizing wounds and drinking water, and later to treat headaches, burns, and itching. |
|||
===Modern period=== |
|||
[[File:AngleseyCopperStream.jpg|right|thumb|[[Acid mine drainage]] affecting the stream running from the disused [[Parys Mountain]] copper mines]] |
|||
The [[Great Copper Mountain]] was a mine in Falun, Sweden, that operated from the 10th century to 1992. It satisfied two thirds of Europe's copper consumption in the 17th century and helped fund many of Sweden's wars during that time.<ref>{{cite book|url = https://books.google.com/books?id=4yp-x3TzDnEC&pg=PA60|page = 60|title = Mining in World History|isbn = 978-1-86189-173-0|author1 = Lynch, Martin|date = 15 April 2004}}</ref> It was referred to as the nation's treasury; Sweden had a [[History of copper currency in Sweden|copper backed currency]].<ref>{{cite web|title=Gold: prices, facts, figures and research: A brief history of money|url=http://www.galmarley.com/FAQs_pages/monetary_history_faqs.htm#Scandinavian%20copper%20money|accessdate=22 April 2011}}</ref> |
|||
Copper is used in roofing,<ref name=":0" /> currency, and for photographic technology known as the [[daguerreotype]]. Copper was used in [[Renaissance]] sculpture, and was used to construct the [[Statue of Liberty]]; copper continues to be used in construction of various types. [[Copper plating]] and [[copper sheathing]] were widely used to protect the under-water hulls of ships, a technique pioneered by the British [[Admiralty]] in the 18th century.<ref>{{cite web|title = Copper and Brass in Ships|url = https://www.copper.org/education/history/60centuries/industrial_age/copperand.html|accessdate = 6 September 2016}}</ref> The [[Norddeutsche Affinerie]] in Hamburg was the first modern [[electroplating]] plant, starting its production in 1876.<ref>{{cite journal|doi = 10.1002/adem.200400403|title = Process Optimization in Copper Electrorefining|date = 2004|author = Stelter, M.|journal = Advanced Engineering Materials|volume = 6|issue = 7|pages=558–562|last2 = Bombach|first2 = H.}}</ref> The German scientist [[Gottfried Osann]] invented [[powder metallurgy]] in 1830 while determining the metal's atomic mass; around then it was discovered that the amount and type of alloying element (e.g., tin) to copper would affect bell tones. [[Flash smelting]] was developed by [[Outokumpu]] in Finland and first applied at [[Harjavalta]] in 1949; the energy-efficient process accounts for 50% of the world's primary copper production.<ref>{{cite web|url = http://www.outokumpu.com/files/Technology/Documents/Newlogobrochures/FlashSmelting.pdf|archiveurl = https://web.archive.org/web/20110724043222/http://www.outokumpu.com/files/Technology/Documents/Newlogobrochures/FlashSmelting.pdf|archivedate = 24 July 2011|title = Outokumpu Flash Smelting|publisher = [[Outokumpu]]|page = 2}}</ref> |
|||
The [[Intergovernmental Council of Copper Exporting Countries]], formed in 1967 by Chile, Peru, Zaire and Zambia, operated in the copper market as [[OPEC]] does in oil, though it never achieved the same influence, particularly because the second-largest producer, the United States, was never a member; it was dissolved in 1988.<ref>{{cite journal |author=Karen A. Mingst |date=1976 |title=Cooperation or illusion: an examination of the intergovernmental council of copper exporting countries |journal=International Organization |volume=30 |issue=2 |pages=263–287 |doi=10.1017/S0020818300018270}}</ref> |
|||
==Applications== |
|||
{{See also| Copper in renewable energy}} |
|||
[[File:Kupferfittings 4062.jpg|thumb|Assorted copper fittings]] |
|||
The major applications of copper are electrical wire (60%), roofing and plumbing (20%), and industrial machinery (15%). Copper is used mostly as a pure metal, but when greater hardness is required, it is put into such alloys as [[brass]] and [[bronze]] (5% of total use).<ref name=emsley/> For more than two centuries, copper paint has been used on boat hulls to control the growth of plants and shellfish.<ref>{{Cite web|url=http://www.boatus.com/magazine/2012/february/copper.asp|title=Is Copper Bottom Paint Sinking?|website=BoatUS Magazine|author=Ryck Lydecker|access-date=2016-06-03}}</ref> A small part of the copper supply is used for nutritional supplements and fungicides in agriculture.<ref name="Boux"/><ref name="Applications for Copper">{{cite web|title = Copper|publisher = [[American Elements]]|date = 2008|url = http://www.americanelements.com/cu.html|accessdate = 12 July 2008}}</ref> [[Machining]] of copper is possible, although alloys are preferred for good [[machinability]] in creating intricate parts. |
|||
===Wire and cable=== |
|||
{{Main article| Copper wire and cable}} |
|||
Despite competition from other materials, copper remains the preferred [[electrical conductor]] in nearly all categories of electrical wiring except overhead [[electric power transmission]] where [[aluminium]] is often preferred.<ref>Pops, Horace, 2008, "Processing of wire from antiquity to the future", ''Wire Journal International'', June, pp. 58–66</ref><ref>The Metallurgy of Copper Wire, http://www.litz-wire.com/pdf%20files/Metallurgy_Copper_Wire.pdf</ref> Copper wire is used in [[power generation]], [[power transmission]], [[power distribution]], [[telecommunications]], [[electronics]] circuitry, and countless types of [[electrical equipment]].<ref>Joseph, Günter, 1999, Copper: Its Trade, Manufacture, Use, and Environmental Status, edited by Kundig, Konrad J.A., ASM International, pps. 141–192 and pps. 331–375.</ref> [[Electrical wiring]] is the most important market for the copper industry.<ref>{{cite web|url=http://www.chemistryexplained.com/elements/C-K/Copper.html |title=Copper, Chemical Element – Overview, Discovery and naming, Physical properties, Chemical properties, Occurrence in nature, Isotopes |publisher=Chemistryexplained.com |accessdate=16 October 2012}}</ref> This includes structural power wiring, power distribution cable, appliance wire, communications cable, automotive wire and cable, and magnet wire. Roughly half of all copper mined is used for electrical wire and cable conductors.<ref>Joseph, Günter, 1999, Copper: Its Trade, Manufacture, Use, and Environmental Status, edited by Kundig, Konrad J.A., ASM International, p.348</ref> Many electrical devices rely on copper wiring because of its multitude of inherent beneficial properties, such as its high [[electrical conductivity]], [[tensile strength]], [[ductility]], [[creep (deformation)]] resistance, [[corrosion]] resistance, low [[thermal expansion]], high [[thermal conductivity]], ease of [[solder]]ing, [[malleability]], and ease of installation. |
|||
For a short period from the late 1960s to the late 1970s, copper wiring was replaced by [[aluminium wiring]] in many housing construction projects in America. The new wiring was implicated in a number of house fires and the industry returned to copper.<ref>{{Cite web|url=https://www.heimer.com/Inspection-Information/Aluminum-Wiring.html|title=Aluminum Wiring Hazards and Pre-Purchase Inspections.|website=www.heimer.com|access-date=2016-06-03}}</ref><ref>{{Cite web|url=http://www.faqs.org/faqs/electrical-wiring/part2/section-16.html|title=Electrical Wiring FAQ (Part 2 of 2)Section - Aluminum wiring|website=www.faqs.org|access-date=2016-06-03}}</ref> |
|||
===Electronics and related devices=== |
|||
[[File:Busbars.jpg|thumb|left|Copper electrical [[busbar]]s distributing power to a large building]] |
|||
[[Integrated circuit]]s and [[printed circuit board]]s increasingly feature copper in place of aluminium because of its superior electrical conductivity; [[heat sink]]s and [[heat exchanger]]s use copper because of its superior heat dissipation properties. [[Electromagnet]]s, [[vacuum tube]]s, [[cathode ray tube]]s, and [[magnetron]]s in microwave ovens use copper, as do [[waveguide]]s for microwave radiation.<ref>{{cite web|title=Accelerator: Waveguides (SLAC VVC)|url=http://www2.slac.stanford.edu/vvc/accelerators/waveguide.html|work=SLAC Virtual Visitor Center|accessdate=29 April 2011}}</ref> |
|||
===Electric motors=== |
|||
Copper's superior [[Copper wire and cable#Electrical conductivity|conductivity]] enhances the efficiency of electrical [[motor (device)|motors]].<ref>IE3 energy-saving motors, Engineer Live, http://www.engineerlive.com/Design-Engineer/Motors_and_Drives/IE3_energy-saving_motors/22687/</ref> This is important because motors and motor-driven systems account for 43%–46% of all global electricity consumption and 69% of all electricity used by industry.<ref>Energy‐efficiency policy opportunities for electric motor‐driven systems, International Energy Agency, 2011 Working Paper in the Energy Efficiency Series, by Paul Waide and Conrad U. Brunner, OECD/IEA 2011</ref> Increasing the mass and cross section of copper in a [[Inductor|coil]] increases the efficiency of the motor. Copper motor rotors, a new technology designed for motor applications where energy savings are prime design objectives,<ref>Fuchsloch, J. and E.F. Brush, (2007), "Systematic Design Approach for a New Series of Ultra‐NEMA Premium Copper Rotor Motors", in EEMODS 2007 Conference Proceedings, 10–15 June, Beijing.</ref><ref>Copper motor rotor project; Copper Development Association; {{cite web|url=http://www.copper.org/applications/electrical/motor-rotor |title=Archived copy |accessdate=2012-11-07 |deadurl=yes |archiveurl=https://web.archive.org/web/20120313102458/http://www.copper.org/applications/electrical/motor-rotor |archivedate=13 March 2012 |df= }}</ref> are enabling general-purpose [[induction motor]]s to meet and exceed [[National Electrical Manufacturers Association]] (NEMA) [[premium efficiency]] standards.<ref>NEMA Premium Motors, The Association of Electrical Equipment and Medical Imaging Manufacturers; {{cite web|url=http://www.nema.org/gov/energy/efficiency/premium/ |title=Archived copy |accessdate=2009-10-12 |deadurl=yes |archiveurl=https://web.archive.org/web/20100402081307/http://www.nema.org/gov/energy/efficiency/premium/ |archivedate=2 April 2010 |df=dmy }}</ref> |
|||
===Architecture=== |
|||
{{Main article|Copper in architecture}} |
|||
[[File:Minneapolis City Hall.jpg|thumb|Copper roof on the [[Minneapolis City Hall]], coated with [[patina]]]] |
|||
[[File:Copper utensils Jerusalem.jpg|thumb|Old copper utensils in a Jerusalem restaurant]] |
|||
Copper has been used since ancient times as a durable, [[corrosion resistance|corrosion resistant]], and weatherproof architectural material.<ref>Seale, Wayne (2007). The role of copper, brass, and bronze in architecture and design; ''Metal Architecture,'' May 2007</ref><ref>Copper roofing in detail; Copper in Architecture; Copper Development Association, U.K., www.cda.org.uk/arch</ref><ref>Architecture, European Copper Institute; http://eurocopper.org/copper/copper-architecture.html</ref><ref>Kronborg completed; Agency for Palaces and Cultural Properties, København, {{cite web|url=http://www.slke.dk/en/slotteoghaver/slotte/kronborg/kronborgshistorie/kronborgfaerdigbygget.aspx?highlight%3Dcopper |title=Archived copy |accessdate=2012-09-12 |deadurl=yes |archiveurl=https://web.archive.org/web/20121024101854/http://www.slke.dk/en/slotteoghaver/slotte/kronborg/kronborgshistorie/kronborgfaerdigbygget.aspx?highlight=copper |archivedate=24 October 2012 |df=dmy }}</ref> [[Roofing material|Roofs]], [[flashing (weatherproofing)|flashings]], [[rain gutter]]s, [[downspout]]s, [[dome]]s, [[spire]]s, vaults, and [[door]]s have been made from copper for hundreds or thousands of years. Copper's architectural use has been expanded in modern times to include interior and exterior [[Copper in architecture#Wall cladding|wall cladding]], building [[expansion joint]]s, [[RF shielding|radio frequency shielding]], and [[Antimicrobial copper-alloy touch surfaces|antimicrobial]] and decorative indoor products such as attractive handrails, bathroom fixtures, and counter tops. Some of copper's other important benefits as an architectural material include low [[thermal expansion|thermal movement]], light weight, [[lightning rod|lightning protection]], and recyclability. |
|||
The metal's distinctive natural green [[patina]] has long been coveted by architects and designers. The final patina is a particularly durable layer that is highly resistant to atmospheric corrosion, thereby protecting the underlying metal against further weathering.<ref>{{cite web|last = Berg|first = Jan|title = Why did we paint the library's roof?|url = http://www.deforest.lib.wi.us/FAQS.htm|accessdate = 20 September 2007 |archiveurl = https://web.archive.org/web/20070625065039/http://www.deforest.lib.wi.us/FAQS.htm |archivedate = 25 June 2007}}</ref><ref>Architectural considerations; Copper in Architecture Design Handbook, http://www.copper.org/applications/architecture/arch_dhb/fundamentals/arch_considerations.htm</ref><ref>Peters, Larry E. (2004). Preventing corrosion on copper roofing systems; Professional Roofing, October 2004, http://www.professionalroofing.net</ref> It can be a mixture of carbonate and sulfate compounds in various amounts, depending upon environmental conditions such as sulfur-containing acid rain.<ref>Oxidation Reaction: Why is the Statue of Liberty Blue-Green? Engage Students in Engineering; www.EngageEngineering.org; Chun Wu, Ph.D., Mount Marty College; Funded by the National Science Foundation (NSF) under Grant No. 083306. {{cite web|url=http://www.wepanknowledgecenter.org/c/document_library/get_file?folderId%3D517%26name%3DDLFE-2454.pdf |title=Archived copy |accessdate=2013-10-25 |deadurl=yes |archiveurl=https://web.archive.org/web/20131025094519/http://www.wepanknowledgecenter.org/c/document_library/get_file?folderId=517&name=DLFE-2454.pdf |archivedate=25 October 2013 |df=dmy }}</ref><ref>{{cite journal |doi=10.1016/S0010-938X(98)00093-6 |title=The chemistry of copper patination |date=1998 |last1=Fitzgerald |first1=K. P. |last2=Nairn |first2=J. |last3=Atrens |first3=A. |journal=Corrosion Science |volume=40 |issue=12 |pages=2029–50}}</ref><ref>Application Areas: Architecture – Finishes – patina; http://www.copper.org/applications/architecture/finishes.html</ref><ref>Glossary of copper terms, Copper Development Association (UK): {{cite web|url=http://www.copperinfo.co.uk/resources/glossary.shtml |title=Archived copy |accessdate=2012-09-14 |deadurl=yes |archiveurl=https://web.archive.org/web/20120820053020/http://www.copperinfo.co.uk/resources/glossary.shtml |archivedate=20 August 2012 |df= }}</ref> Architectural copper and its alloys can also be [[Copper in architecture#Finishes|'finished']] to embark a particular look, feel, and/or color. Finishes include mechanical surface treatments, chemical coloring, and coatings.<ref>Finishes – natural weathering; Copper in Architecture Design Handbook, Copper Development Association Inc., {{cite web|url=http://www.copper.org/applications/architecture/arch_dhb/finishes/finishes.html |title=Archived copy |accessdate=2012-09-12 |deadurl=yes |archiveurl=https://web.archive.org/web/20121016080539/http://www.copper.org/applications/architecture/arch_dhb/finishes/finishes.html |archivedate=16 October 2012 |df= }}</ref> |
|||
Copper has excellent [[brazing]] and [[soldering]] properties and can be [[welded]]; the best results are obtained with [[gas metal arc welding]].<ref>{{cite book|author = Davis, Joseph R. |title = Copper and Copper Alloys|pages = 3–6, 266|publisher = ASM International|date = 2001|isbn = 0-87170-726-8}}</ref> |
|||
===Antibiofouling applications=== |
|||
{{Main article|Copper alloys in aquaculture|Copper sheathing}} |
|||
Copper is [[biostatic]], meaning bacteria and many other forms of life will not grow on it. For this reason it has long been used to line parts of ships to protect against [[barnacle]]s and [[mussel]]s. It was originally used pure, but has since been superseded by [[Muntz metal]] and copper-based paint. Similarly, as discussed in [[copper alloys in aquaculture]], copper alloys have become important netting materials in the [[aquaculture]] industry because they are [[antimicrobial]] and prevent [[biofouling]], even in extreme conditions<ref name="autogenerated1995">Edding, Mario E., Flores, Hector, and Miranda, Claudio, (1995), Experimental Usage of Copper-Nickel Alloy Mesh in Mariculture. Part 1: Feasibility of usage in a temperate zone; Part 2: Demonstration of usage in a cold zone; Final report to the International Copper Association Ltd.</ref> and have strong structural and [[corrosion-resistant]]<ref>[http://www.copper.org/applications/cuni/pdf/marine_aquaculture.pdf Corrosion Behaviour of Copper Alloys used in Marine Aquaculture]. (PDF) . copper.org. Retrieved on 8 November 2011.</ref> properties in marine environments. |
|||
===Antimicrobial applications=== |
|||
{{Main article|Antimicrobial properties of copper|Antimicrobial copper-alloy touch surfaces}} |
|||
[[Antimicrobial copper-alloy touch surfaces|Copper-alloy touch surfaces]] have natural properties that destroy a wide range of [[microorganisms]] (e.g., ''[[Escherichia coli|E. coli]]'' O157:H7, [[methicillin]]-resistant ''[[Staphylococcus aureus]]'' ([[Methicillin-resistant Staphylococcus aureus|MRSA]]), ''[[Staphylococcus]]'', ''[[Clostridium difficile (bacteria)|Clostridium difficile]]'', [[influenza A virus]], [[Adenoviridae|adenovirus]], and [[Fungus|fungi]]).<ref name="Copper Touch Surfaces">[http://coppertouchsurfaces.org/antimicrobial/bacteria/index.html Copper Touch Surfaces]. Copper Touch Surfaces. Retrieved on 8 November 2011.</ref> Some 355 copper alloys{{clarify|reason=please explain, what is a 355 copper alloy?|date=June 2016}} were proven to kill more than 99.9% of disease-causing bacteria within just two hours when cleaned regularly.<ref name="epa.gov">[http://www.epa.gov/pesticides/factsheets/copper-alloy-products.htm EPA registers copper-containing alloy products], May 2008</ref> The [[United States Environmental Protection Agency]] (EPA) has approved the registrations of these copper alloys as "[[antimicrobial]] materials with public health benefits";<ref name="epa.gov"/> that approval allows manufacturers to make legal claims to the public health benefits of products made of registered alloys. In addition, the EPA has approved a long list of antimicrobial copper products made from these alloys, such as bedrails, [[handrails]], over-bed tables, [[sinks]], [[faucets]], [[door knobs]], [[toilet]] hardware, [[computer keyboards]], [[health club]] equipment, and [[shopping cart]] handles (for a comprehensive list, see: [[Antimicrobial copper-alloy touch surfaces#Approved products]]). Copper doorknobs are used by hospitals to reduce the transfer of disease, and [[Legionnaires' disease]] is suppressed by copper tubing in plumbing systems.<ref>{{cite journal|last1=Biurrun|first1=Amaya|last2=Caballero|first2=Luis|last3=Pelaz|first3=Carmen|last4=León|first4=Elena|last5=Gago|first5=Alberto|title=Treatment of a Legionella pneumophila‐Colonized Water Distribution System Using Copper‐Silver Ionization and Continuous Chlorination|journal=Infection Control and Hospital Epidemiology|date=1999|volume=20|issue=6|pages=426–428|doi=10.1086/501645|jstor=30141645|pmid=10395146}}</ref> Antimicrobial copper alloy products are now being installed in healthcare facilities in the U.K., Ireland, Japan, Korea, France, Denmark, and Brazil{{citation needed|date=November 2013}} and in the subway transit system in Santiago, Chile, where copper-zinc alloy handrails will be installed in some 30 stations between 2011 and 2014.<ref>[http://www.rail.co/2011/07/22/chilean-subway-protected-with-antimicrobial-copper Chilean subway protected with Antimicrobial Copper – Rail News from] {{webarchive|url=https://web.archive.org/web/20120724105812/http://www.rail.co/2011/07/22/chilean-subway-protected-with-antimicrobial-copper/ |date=24 July 2012 }}. rail.co. Retrieved on 8 November 2011.</ref><ref>[http://construpages.com.ve/nl/noticia_nl.php?id_noticia=3032&language=en Codelco to provide antimicrobial copper for new metro lines (Chile)] {{dead link|date=September 2016|bot=medic}}{{cbignore|bot=medic}}. Construpages.com.ve. Retrieved on 8 November 2011.</ref><ref>[http://www.antimicrobialcopper.com/media/149689/pr811-chilean-subway-installs-antimicrobial-copper.pdf PR 811 Chilean Subway Installs Antimicrobial Copper] {{webarchive|url=https://web.archive.org/web/20111123100624/http://www.antimicrobialcopper.com/media/149689/pr811-chilean-subway-installs-antimicrobial-copper.pdf |date=23 November 2011 }}. (PDF). antimicrobialcopper.com. Retrieved on 8 November 2011.</ref> |
|||
===Folk medicine=== |
|||
Copper is commonly used in jewelry, and according to some folklore, copper bracelets relieve [[arthritis]] symptoms.<ref>{{cite journal |pmid=961545 |date=1976 |last1=Walker |first1=W. R. |last2=Keats |first2=D. M. |title=An investigation of the therapeutic value of the 'copper bracelet'-dermal assimilation of copper in arthritic/rheumatoid conditions |volume=6 |issue=4 |pages=454–9 |journal=Agents and actions}}</ref> In various studies, though, no difference is found between arthritis treated with a copper bracelet, magnetic bracelet, or placebo bracelet.<ref>{{cite journal | pmid = 19942103 | doi=10.1016/j.ctim.2009.07.002 | volume=17 | title=Therapeutic effects of magnetic and copper bracelets in osteoarthritis: a randomised placebo-controlled crossover trial | year=2009 | journal=Complement Ther Med | pages=249–56 | last1 = Richmond | first1 = SJ | last2 = Brown | first2 = SR | last3 = Campion | first3 = PD | display-authors = 3 | last4 = et al}}<br /><br />No difference was observed between devices in terms of their effects on pain as measured by the primary outcome measure (WOMAC A), the PRI and the VAS. Similar results were obtained for stiffness (WOMAC B), physical function (WOMAC C), and medication use. Further analyses of the PRI subscales revealed a statistically significant difference between devices (P=0.025), which favoured the experimental device. Participants reported lower sensory pain after wearing the standard magnetic wrist strap, than when wearing control devices. However, no adjustment was made for multiple testing.</ref><ref name="RichmondBrown2009">{{cite journal|last1=Richmond|first1=Stewart J.|last2=Brown|first2=Sally R.|last3=Campion|first3=Peter D.|last4=Porter|first4=Amanda J. L.|last5=Moffett|first5=Jennifer A. Klaber|last6=Jackson|first6=David A.|last7=Featherstone|first7=Valerie A.|last8=Taylor|first8=Andrew J.|title=Therapeutic effects of magnetic and copper bracelets in osteoarthritis: A randomised placebo-controlled crossover trial|journal=Complementary Therapies in Medicine|volume=17|issue=5–6|year=2009|pages=249–256|issn=0965-2299|doi=10.1016/j.ctim.2009.07.002|pmid=19942103}}</ref> Medical science has not demonstrated any benefits in copper jewelry for any medical condition. A human being can have a dietary copper deficiency, but the condition is very rare because copper is present in many common foods, including [[legumes]] (beans), grains, and [[nut (fruit)|nut]]s.<ref>University of Arkansas for Medical Sciences:<br />[http://www.uamshealth.com/?id=882&sid=1 Can wearing a copper bracelet cure arthritis?]<br /><br />According to the Center for Hand and Upper Extremity Surgery at UAMS, copper deficiency is extremely rare and most regular diets provide enough copper to meet the daily requirements. Copper is a component of some of the normal cellular enzymes in most mineral rich foods, such as vegetables, potatoes, legumes (beans and peas), nuts (peanuts and pecans), grains (wheat and rye) and fruits. Supplementation is only needed in patients with serious medical conditions that affect their gastrointestinal tract and impair their ability to absorb nutrients. |
|||
</ref> |
|||
No evidence shows that copper can be absorbed through the skin. If it were, it might lead to [[Copper toxicity|copper poisoning]].<ref>University of Arkansas for Medical Sciences:<br />[http://www.uams.edu/update/absolutenm/templates/medical.asp?articleid=3454 Find the Truth Behind Medical Myths] {{webarchive |url=https://web.archive.org/web/20140106233901/http://www.uams.edu/update/absolutenm/templates/medical.asp?articleid=3454 |date=6 January 2014 }}<br /><br />While it's never been proven that copper can copper be absorbed through the skin by wearing a bracelet, research has shown that excessive copper can result in poisoning, causing vomiting and, in severe cases, liver damage.</ref> |
|||
====Compression clothing==== |
|||
Recently, some [[compression bandage|compression]] clothing with inter-woven copper has been marketed with the same [[traditional medicine|folk medicine]] claims. Because compression clothing is a valid treatment for some ailments, the clothing may appear to work, but the added copper may have no benefit beyond a [[placebo effect]].<ref> |
|||
Truth in Advertising<br />[https://www.truthinadvertising.org/tommie-copper/ Tommie Copper]<br /><br />So it seems possible that copper-infused compression clothing could help you recover from a tough workout, and it's also possible it could have some anti-bacterial properties in clothes. But as for the claims in the infomercial about relieving joint pain and helping with everyday aches — any relief from copper-compression seems more likely to be a placebo effect than anything else. Think carefully before shelling out for Tommie Copper. |
|||
</ref> |
|||
===Other uses=== |
|||
{{Repetition section|date=June 2017}} |
|||
Solutions of copper compounds are used as a wood preservative, particularly in treating the original portion of structures during restoration of [[dry rot]] damage. Together with zinc, copper wires may be installed over non-conductive roofing materials to discourage the growth of moss.{{citation needed|date=November 2013}} Textile fibers are blended with copper to create antimicrobial protective fabrics.<ref>{{cite web |title= Copper and Cupron |publisher=Cupron |url=http://www.cupron.com/cupron-technology/power-of-cupron/copper-and-cupron}}{{unreliable source?|date=November 2013}}</ref><ref name="Ergowear">[https://www.ergowear.com/incopper-specifications/ Ergowear], Copper antimicrobial yarn technology used in male underwear{{unreliable source?|date=November 2013}}</ref> Copper alloys are used in [[musical instrument]]s, particularly: the body of [[brass instrument]]s; circuitry for all those that are electronically amplified; the bodies of brass percussion such as [[gong]]s, [[bell]]s, and [[kettle drums]]; tuning heads on [[guitar]]s and other [[string instrument]]s; string windings on harps, pianos, [[harpsichord]]s, and string instruments; and the frame elements of [[piano]]s and [[harp]]s. Copper is commonly used as a base on which other metals such as nickel are electroplated. |
|||
Copper is one of three metals, along with lead and silver, used in the museum materials testing procedure called the [[Oddy test]] to detect chlorides, oxides, and sulfur compounds. |
|||
Copper is used as the printing plate in [[etching]], [[engraving]] and other forms of [[intaglio (printmaking)|intaglio]] [[printmaking]]. |
|||
Copper oxide and carbonate are used add color in [[stained glass|stain glass]] works, in [[glass]]making, and in [[ceramic glazes]] to impart turquoise blue, green, and brown colors. |
|||
Copper is used to create stills for distilling spirits, for example to make [[whisky]]. It's malleability makes it easy to bend into the various shapes required and allows considerable flexibility in the shaping of the still and associated pipework; the metal also reacts with undesirable sulfur-containing components in the vapor and distillate making for a cleaner product. |
|||
==Degradation== |
|||
''[[Chromobacterium violaceum]]'' and ''[[Pseudomonas fluorescens]]'' can both mobilize solid copper as a cyanide compound.<ref name="Geoffrey Michael Gadd 609–643">{{cite journal|url=http://mic.sgmjournals.org/content/156/3/609.full|title=Metals, minerals and microbes: geomicrobiology and bioremediation|journal=Microbiology|author=Geoffrey Michael Gadd|volume=156|issue=3|date=March 2010|pages=609–643|doi=10.1099/mic.0.037143-0|pmid=20019082}}</ref> The ericoid mycorrhizal fungi associated with ''Calluna'', ''Erica'' and ''Vaccinium'' can grow in metalliferous soils containing copper.<ref name="Geoffrey Michael Gadd 609–643"/> The ectomycorrhizal fungus ''Suillus luteus'' protects young pine trees from copper toxicity. A sample of the fungus ''[[Aspergillus niger]]'' was found growing from gold mining solution and was found to contain cyano complexes of such metals as gold, silver, copper, iron, and zinc. The fungus also plays a role in the solubilization of heavy metal sulfides.<ref>{{cite book|url=https://books.google.com/?id=WY3YvfNoouMC&pg=PA533#v=onepage&q&f=false|title=Mycoremediation: Fungal Bioremediation|author=Harbhajan Singh|page=509|isbn=978-0-470-05058-3|date=2006-11-17}}</ref> |
|||
==Biological role== |
|||
{{Main article|Copper in health}} |
|||
[[File:ARS copper rich foods.jpg|thumb|Rich sources of copper include oysters, beef and lamb liver, Brazil nuts, blackstrap molasses, cocoa, and black pepper. Good sources include lobster, nuts and sunflower seeds, green olives, avocados, and wheat bran.]] |
|||
[[Copper proteins]] have diverse roles in biological electron transport and oxygen transportation, processes that exploit the easy interconversion of Cu(I) and Cu(II).<ref>{{cite book |
|||
|first1=Katherine E. |last1=Vest|first2=Hayaa F.|last2=Hashemi|first3=Paul A.|last3=Cobine |
|||
|editor1-first=Lucia |editor1-last=Banci |series=Metal Ions in Life Sciences |volume=12 |
|||
|chapter= Chapter 13 The Copper Metallome in Eukaryotic Cells |
|||
|title=Metallomics and the Cell |date=2013 |publisher=Springer |isbn=978-94-007-5560-4|doi=10.1007/978-94-007-5561-10_12}} electronic-book {{ISBN|978-94-007-5561-1}} {{ISSN|1559-0836}} electronic-{{ISSN|1868-0402}} |
|||
</ref><ref>{{cite book |
|||
|first1=Katherine E. |last1=Vest|first2=Hayaa F.|last2=Hashemi |first3=Paul A.|last3=Cobine |
|||
|editor1-first=Lucia |editor1-last=Banci |series=Metal Ions in Life Sciences |volume=12 |
|||
|chapter= Chapter 12 The Copper Metallome in Prokaryotic Cells |
|||
|title=Metallomics and the Cell |date=2013 |publisher=Springer |isbn=978-94-007-5560-4|doi=10.1007/978-94-007-5561-10_13}} electronic-book {{ISBN|978-94-007-5561-1}} {{ISSN|1559-0836}} electronic-{{ISSN|1868-0402}} |
|||
</ref><ref> |
|||
{{cite book |
|||
|first1=Gereon M. |
|||
|last1=Yee |
|||
|first2=William B. |
|||
|last2=Tolman |
|||
|editor=Peter M.H. Kroneck and Martha E. Sosa Torres |
|||
|title=Sustaining Life on Planet Earth: Metalloenzymes Mastering Dioxygen and Other Chewy Gases |
|||
|series=Metal Ions in Life Sciences |
|||
|volume=15 |
|||
|year=2015 |
|||
|publisher=Springer |
|||
|chapter=Chapter 5, Section 4 ''Dioxygen Activation by Copper Complexes'' |
|||
|pages=175–192 |
|||
|doi=10.1007/978-3-319-12415-5_5 |
|||
}} |
|||
</ref> |
|||
<ref name="Lippard">S. J. Lippard, J. M. Berg "Principles of bioinorganic chemistry" University Science Books: Mill Valley, CA; 1994. {{ISBN|0-935702-73-3}}.</ref> The biological role for copper commenced with the appearance of oxygen in earth's atmosphere.<ref>{{cite journal|pmid=10821735|author=Decker, H.|author2=Terwilliger, N.|last-author-amp=yes |title=COPs and Robbers: Putative evolution of copper oxygen-binding proteins|journal= Journal of Experimental Biology |volume=203|pages=1777–1782 |date=2000|issue=Pt 12}}</ref> |
|||
Copper is essential in the aerobic [[Cellular respiration|respiration]] of all [[eukaryotes]]. In [[mitochondria]], it is found in [[cytochrome c oxidase]], which is the last protein in [[oxidative phosphorylation]]. Cytochrome c oxidase is the protein that binds the O<sub>2</sub> between a copper and an iron; the protein transfers 8 electrons to the O<sub>2</sub> molecule to reduce it to two molecules of water. |
|||
Copper is also found in many [[superoxide dismutase]]s, proteins that catalyze the decomposition of [[superoxide]]s by converting it (by [[disproportionation]]) to oxygen and [[hydrogen peroxide]]: |
|||
:2 HO<sub>2</sub> → H<sub>2</sub>O<sub>2</sub> + O<sub>2</sub> |
|||
The protein [[hemocyanin]] is the oxygen carrier in most [[mollusk]]s and some [[arthropod]]s such as the [[horseshoe crab]] (''Limulus polyphemus'').<ref name=NOAA>{{cite web|title = Fun facts|work = Horseshoe crab|publisher = University of Delaware|url = http://www.ocean.udel.edu/horseshoecrab/funFacts.html|accessdate = 13 July 2008}}</ref> Because hemocyanin is blue, these organisms have blue blood rather than the red blood of iron-based [[hemoglobin]]. Structurally related to hemocyanin are the [[laccase]]s and [[tyrosinase]]s. Instead of reversibly binding oxygen, these proteins hydroxylate substrates, illustrated by their role in the formation of [[lacquer]]s.<ref name="Lippard"/> |
|||
Several copper proteins, such as the "blue copper proteins", do not interact directly with substrates, hence they are not enzymes. These proteins relay electrons by the process called [[electron transfer]].<ref name="Lippard"/> |
|||
[[File:Thylakoid membrane.png|thumb|440px|Photosynthesis functions by an elaborate electron transport chain within the [[thylakoid membrane]]. A central link in this chain is [[plastocyanin]], a blue copper protein.]] |
|||
A unique tetranuclear copper center has been found in [[nitrous-oxide reductase]].<ref> |
|||
{{cite book |
|||
|first1=Lisa K. |
|||
|last1= Schneider |
|||
|first2=Anja |
|||
|last2= Wüst |
|||
|first3=Anja |
|||
|last3= Pomowski |
|||
|first4=Lin |
|||
|last4= Zhang |
|||
|first5=Oliver |
|||
|last5= Einsle |
|||
|editor=Peter M.H. Kroneck |
|||
|editor2=Martha E. Sosa Torres |
|||
|title=The Metal-Driven Biogeochemistry of Gaseous Compounds in the Environment |
|||
|series=Metal Ions in Life Sciences |
|||
|volume=14 |
|||
|date=2014 |
|||
|publisher=Springer |
|||
|chapter=Chapter 8. ''No Laughing Matter: The Unmaking of the Greenhouse Gas Dinitrogen Monoxide by Nitrous Oxide Reductase'' |
|||
|pages=177–210 |
|||
|doi=10.1007/978-94-017-9269-1_8 |
|||
}} |
|||
</ref> |
|||
===Dietary needs=== |
|||
Copper is an essential [[trace element]] in plants and animals, but not all microorganisms. The human body contains copper at a level of about 1.4 to 2.1 mg per kg of body mass.<ref name="copper.org">{{cite web|url = http://www.copper.org/consumers/health/papers/cu_health_uk/cu_health_uk.html|title = Amount of copper in the normal human body, and other nutritional copper facts|accessdate = 3 April 2009}}</ref> Copper is absorbed in the gut, then transported to the liver bound to [[serum albumin|albumin]].<ref>{{cite journal|last1=Adelstein|first1=S. J.|last2=Vallee|first2=B. L.|title=Copper metabolism in man|journal=New England Journal of Medicine|date=1961|volume=265|pages=892–897|doi=10.1056/NEJM196111022651806|issue=18}}</ref> After processing in the liver, copper is distributed to other tissues in a second phase, which involves the protein [[ceruloplasmin]], carrying the majority of copper in blood. Ceruloplasmin also carries the copper that is excreted in milk, and is particularly well-absorbed as a copper source.<ref>{{cite journal | url = http://www.ajcn.org/content/67/5/965S.abstract | title = Copper transport | pmid = 9587137 | date = 1 May 1998 | author1 = M C Linder | journal = The American Journal of Clinical Nutrition | volume = 67 | issue = 5 | pages = 965S–971S | last2 = Wooten | first2 = L. | last3 = Cerveza | first3 = P. | last4 = Cotton | first4 = S. | last5 = Shulze | first5 = R. | last6 = Lomeli | first6 = N.}}</ref> Copper in the body normally undergoes [[enterohepatic circulation]] (about 5 mg a day, vs. about 1 mg per day absorbed in the diet and excreted from the body), and the body is able to excrete some excess copper, if needed, via [[bile]], which carries some copper out of the liver that is not then reabsorbed by the intestine.<ref>{{cite journal | jstor =20170553 | pmid = 775938 | date =1976 | last1 =Frieden | first1 =E. | last2 =Hsieh | first2 =H. S. | title =Ceruloplasmin: The copper transport protein with essential oxidase activity | volume =44 | pages =187–236 | journal =Advances in Enzymology and Related Areas of Molecular Biology | doi=10.1002/9780470122891.ch6| series = Advances in Enzymology - and Related Areas of Molecular Biology | isbn = 978-0-470-12289-1}}</ref><ref>{{cite journal | url =http://ajpcell.physiology.org/content/258/1/C140 | pmid =2301561 | title =Copper transport from ceruloplasmin: Characterization of the cellular uptake mechanism | date =1 January 1990 | author1 =S. S. Percival | journal = American Journal of Physiology. Cell Physiology | volume =258 | issue =1 | pages =C140–6 | last2 =Harris | first2 =E. D.}}</ref> |
|||
===Dietary reference intake=== |
|||
The Food and Nutrition Board of the U.S. Institute of Medicine updated the estimated average requirements (EARs) and recommended dietary allowances (RDAs) for copper in 2001. The current EAR for copper for people age 14 and older is 0.7 mg/day. The RDA is 0.9 mg/day. RDAs are higher than EARs so as to identify amounts that will cover people with higher than average requirements. The RDA for pregnant women equals 1.0 mg/day. The RDA for lactating women equals 1.3 mg/day. For infants up to 12 months the AI is 0.22 mg/day and for children age 1–13 years the RDA increases with age from 0.34 to 0.7 mg/day. The Food and Nutrition Board also sets tolerable upper intake Levels (ULs) for vitamins and minerals when evidence is sufficient. In the case of copper the UL is set at 10 mg/day. Collectively the EARs, RDAs, AIs and ULs are referred to as [[Dietary Reference Intake]]s.<ref>Copper. IN: [https://www.nap.edu/read/10026/chapter/9 Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Copper]. National Academy Press. 2001, PP. 224–257.</ref> The European Food Safety Authority reviewed the same safety question and set its UL at 5 mg/day.<ref>{{citation| title = Tolerable Upper Intake Levels For Vitamins And Minerals| publisher = European Food Safety Authority| year = 2006| url = http://www.efsa.europa.eu/sites/default/files/efsa_rep/blobserver_assets/ndatolerableuil.pdf}}</ref> |
|||
For U.S. food and dietary supplement labeling purposes the amount in a serving is expressed as a percent of daily value (%DV). For copper labeling purposes 100% of the DV was 2.0 mg, but as of May 2016 it has been revised to 0.9 mg. Food and supplement companies have until July 28, 2018 to comply with the change. |
|||
===Copper-based disorders=== |
|||
Because of its role in facilitating iron uptake, [[copper deficiency]] can produce [[anemia]]-like symptoms, [[neutropenia]], bone abnormalities, hypopigmentation, impaired growth, increased incidence of infections, osteoporosis, hyperthyroidism, and abnormalities in glucose and cholesterol metabolism. Conversely, [[Wilson's disease]] causes an accumulation of copper in body tissues. |
|||
Severe deficiency can be found by testing for low plasma or serum copper levels, low ceruloplasmin, and low red blood cell superoxide dismutase levels; these are not sensitive to marginal copper status. The "cytochrome c oxidase activity of leucocytes and platelets" has been stated as another factor in deficiency, but the results have not been confirmed by replication.<ref name=Bonhametal2002>{{cite journal|last=Bonham |first= Maxine |last2= O'Connor |first2= Jacqueline M. |last3= Hannigan |first3= Bernadette M. |last4= Strain |first4= J. J.|date=2002|title=The immune system as a physiological indicator of marginal copper status? |journal=British Journal of Nutrition|doi=10.1079/BJN2002558|pmid=12010579|volume=87|issue=5|pages=393–403}}</ref> |
|||
{{NFPA 704|Health = 2|Flammability = 0|Reactivity = 0|S=|caption=Fire diamond for copper metal}} |
|||
{{Main article|Copper toxicity}} |
|||
Gram quantities of various copper salts have been taken in suicide attempts and produced acute copper toxicity in humans, possibly due to redox cycling and the generation of [[reactive oxygen species]] that damage [[DNA]].<ref>{{cite journal|last1=Li|first1=Yunbo|last2=Trush|first2=Michael|last3=Yager|first3=James|title=DNA damage caused by reactive oxygen species originating from a copper-dependent oxidation of the 2-hydroxy catechol of estradiol|journal=Carcinogenesis|date=1994|volume=15|issue=7|pages=1421–1427|doi=10.1093/carcin/15.7.1421|pmid=8033320}}</ref><ref>{{cite journal|last1=Gordon|first1=Starkebaum|last2=John|first2=M. Harlan|title=Endothelial cell injury due to copper-catalyzed hydrogen peroxide generation from homocysteine|pmc=424498|pmid=3514679|doi=10.1172/JCI112442|volume=77|issue=4|date=April 1986|journal=J. Clin. Invest.|pages=1370–6}}</ref> Corresponding amounts of copper salts (30 mg/kg) are toxic in animals.<ref>{{cite web|title = Pesticide Information Profile for Copper Sulfate|url = http://pmep.cce.cornell.edu/profiles/extoxnet/carbaryl-dicrotophos/copper-sulfate-ext.html|publisher = Cornell University|accessdate=10 July 2008}}</ref> A minimum dietary value for healthy growth in rabbits has been reported to be at least 3 [[Parts per million|ppm]] in the diet.<ref>{{cite journal|author=Hunt, Charles E.|author2=William W. Carlton|last-author-amp=yes |pmid=5841854 |date=1965|title=Cardiovascular Lesions Associated with Experimental Copper Deficiency in the Rabbit|journal=Journal of Nutrition |volume=87|pages=385–394|issue=4}}</ref> However, higher concentrations of copper (100 ppm, 200 ppm, or 500 ppm) in the diet of rabbits may favorably influence [[Feed conversion ratio|feed conversion efficiency]], growth rates, and carcass dressing percentages.<ref>{{cite journal|url=http://riunet.upv.es/handle/10251/10503?locale-attribute=en|author=Ayyat M.S.|author2=Marai I.F.M.|author3=Alazab A.M. |date=1995|title=Copper-Protein Nutrition of New Zealand White Rabbits under Egyptian Conditions|journal= World Rabbit Science |volume=3|issue=3 |pages=113–118|doi=10.4995/wrs.1995.249}}</ref> |
|||
Chronic copper toxicity does not normally occur in humans because of transport systems that regulate absorption and excretion. Autosomal recessive mutations in copper transport proteins can disable these systems, leading to [[Wilson's disease]] with copper accumulation and [[cirrhosis]] of the liver in persons who have inherited two defective genes.<ref name="copper.org"/> |
|||
Elevated copper levels have also been linked to worsening symptoms of [[Alzheimer's disease]].<ref>{{vcite journal | author = Brewer GJ | title = Copper excess, zinc deficiency, and cognition loss in Alzheimer's disease | journal = BioFactors (Oxford, England) | volume = 38 | issue = 2 | pages = 107–113 | date = March 2012 | pmid = 22438177 | doi = 10.1002/biof.1005 | type = Review}}</ref><ref>{{cite web|title=Copper: Alzheimer's Disease|url=http://examine.com/supplements/Copper#summary9-0|publisher=[[Examine.com]]|accessdate=21 June 2015}}</ref> |
|||
=== Human exposure === |
|||
In the US, the [[Occupational Safety and Health Administration]] (OSHA) has designated a [[permissible exposure limit]] (PEL) for copper dust and fumes in the workplace as a time-weighted average (TWA) of 1 mg/m<sup href="Legionnaires' disease">3</sup>. The [[National Institute for Occupational Safety and Health]] (NIOSH) has set a [[Recommended exposure limit]] (REL) of 1 mg/m<sup>3</sup>, time-weighted average. The [[IDLH]] (immediately dangerous to life and health) value is 100 mg/m<sup href="arthritis">3</sup>.<ref>{{PGCH|0150}}</ref> |
|||
Copper is a constituent of [[tobacco smoke]].<ref>[[OEHHA]] ''[https://oehha.ca.gov/chemicals/copper Copper]''</ref><ref name="TalhoutSchulz2011">{{cite journal|last1=Talhout|first1=Reinskje|last2=Schulz|first2=Thomas|last3=Florek|first3=Ewa|last4=Van Benthem|first4=Jan|last5=Wester|first5=Piet|last6=Opperhuizen|first6=Antoon|title=Hazardous Compounds in Tobacco Smoke|journal=International Journal of Environmental Research and Public Health|volume=8|issue=12|year=2011|pages=613–628|issn=1660-4601|doi=10.3390/ijerph8020613|pmid=21556207|pmc=3084482}}</ref> The [[tobacco plant]] readily absorbs and accumulates [[heavy metals]], such as copper from the surrounding soil into its leaves. These are readily absorbed into the user's body following smoke inhalation.<ref>Alireza Pourkhabbaz, Hamidreza Pourkhabbaz [http://www.sciencedirect.com/science/article/pii/S0039914011010952 Investigation of Toxic Metals in the Tobacco of Different Iranian Cigarette Brands and Related Health Issues], Iran J Basic Med Sci. 2012 Jan-Feb; 15(1): 636–644. {{PMC|3586865}}</ref> The health implications are not clear.<ref>David Bernhard, Andrea Rossmann, and Georg Wick [http://onlinelibrary.wiley.com/doi/10.1080/15216540500459667/pdf Metals in Cigarette Smoke], IUBMB Life, 57(12): 805–809, December 2005</ref> |
|||
==See also== |
|||
* [[Erosion corrosion of copper water tubes]] |
|||
** [[Cold water pitting of copper tube]] |
|||
* [[List of countries by copper production]] |
|||
* [[Metal theft]] |
|||
** [[Operation Tremor]] |
|||
* [[Smelter]] |
|||
* [[Peak copper]] |
|||
** [[Anaconda Copper]] |
|||
** [[Antofagasta PLC]] |
|||
** [[Bingham Canyon Mine]] |
|||
** [[Codelco]] |
|||
** [[Grasberg mine]] |
|||
** [[El Boleo|El Boleo mine]] |
|||
==References== |
|||
{{Reflist|colwidth=30em}} |
|||
==Notes== |
|||
{| Class = "wikitable" style = "text-align: center" |
|||
|+[[Pourbaix diagram]]s for copper |
|||
<!--| style="width:25px;"|[[File:Copper in water pourbiax diagram.png|center|200px]] |
|||
| style="width:25px;"|[[File:Copper in sulphide media pourbiax diagram.png|center|200px]] |
|||
| style="width:25px;"|[[File:Copper in 10M ammonia pourbiax diagram.png|center|200px]] |
|||
| style="width:25px;"|[[File:Copper in chloride media more copper pourbiax.png|center|200px]]--> |
|||
|- |
|||
|in pure water, or acidic or alkali conditions. Copper in neutral water is more noble than hydrogen. |
|||
|in water containing sulfide |
|||
|in 10 M ammonia solution |
|||
|in a chloride solution |
|||
|} |
|||
==Further reading== |
|||
* {{cite book|title=Handbook of Copper Pharmacology and Toxicology|editor=Massaro, Edward J.|publisher=Humana Press|date=2002|isbn=0-89603-943-9}} |
|||
* {{cite web|title=''Copper: Technology & Competitiveness (Summary)'' Chapter 6: Copper Production Technology|publisher=Office of Technology Assessment|date=2005|url=http://www.princeton.edu/~ota/disk2/1988/8808/880808.PDF}} |
|||
* Current Medicinal Chemistry, Volume 12, Number 10, May 2005, pp. 1161–1208(48) Metals, Toxicity and Oxidative Stress |
|||
* {{cite book|title=Materials Science and Engineering: an Introduction|edition=6th|author=William D. Callister|publisher=Wiley, New York|date=2003|isbn=0-471-73696-1|at=Table 6.1, p. 137}} |
|||
* [http://www.memsnet.org/material/coppercubulk/ Material: Copper (Cu), bulk], MEMS and Nanotechnology Clearinghouse. |
|||
* {{cite journal|author=Kim BE|author2= Nevitt T|author3=Thiele DJ|title=Mechanisms for copper acquisition, distribution and regulation|journal=Nat. Chem. Biol.|volume=4|date=2008|pmid=18277979|doi=10.1038/nchembio.72|url=http://www.nature.com/nchembio/journal/v4/n3/abs/nchembio.72.html|issue=3|pages=176–85}} |
|||
* [http://www.rsc.org/Publishing/Journals/cb/Volume/2009/1/Copper.asp Copper transport disorders]: an Instant insight from the [[Royal Society of Chemistry]] |
|||
==External links== |
|||
{{Wikiquote}} |
|||
{{Commons|Copper}} |
|||
{{Wiktionary|copper}} |
|||
{{Wikisource}} |
|||
* [http://www.periodicvideos.com/videos/029.htm Copper] at ''[[The Periodic Table of Videos]]'' (University of Nottingham) |
|||
* [http://www.npi.gov.au/substances/copper/index.html Copper and compounds fact sheet] from the [[National Pollutant Inventory]] of Australia |
|||
* [http://www.cdc.gov/niosh/npg/npgd0150.html CDC–NIOSH Pocket Guide to Chemical Hazards – Copper (dusts and mists)] from the [[Centers for Disease Control and Prevention]]'s [[National Institute for Occupational Safety and Health]] |
|||
* [http://www.cdc.gov/niosh/npg/npgd0151.html CDC–NIOSH Pocket Guide to Chemical Hazards – Copper fume] from the [[Centers for Disease Control and Prevention]]'s [[National Institute for Occupational Safety and Health]] |
|||
* [http://www.copper.org Copper.org] - official website of the Copper Development Association with an extensive site of properties and uses of copper |
|||
** [http://www.brass.org Brass.org] - also operated by the Copper Development Association; dedicated to [[brass]], a copper alloy. |
|||
* [http://www.indexmundi.com/commodities/?commodity=copper&months=300 Price history of copper, according to the IMF] |
|||
{{compact periodic table}} |
|||
{{Copper compounds}} |
|||
{{Jewellery}} |
|||
<!--{{Authority control}} |
|||
[[Kategori:Tembaga| ]] |
|||
[[Kategori:Unsur kimia]] |
|||
[[Kategori:Logam transisi]] |
|||
[[Kategori:Biologi dan farmakologi unsur kimia]] |
|||
[[Kategori:Mineral diet]] |
|||
[[Kategori:Penghantar listrik]] |
|||
[[Kategori:Perusahaan pertambangan tembaga]] |
|||
[[Kategori:Mineral kubik]] |
|||
[[Kategori:Mineral unsur asli]] |
|||
--> |
|||
Revisi terkini sejak 23 Juli 2020 10.14
Templat:Infobox copper Copper is a chemical element with symbol Cu (from bahasa Latin: cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a reddish-orange color. Copper is used as a conductor of heat and electricity, as a building material, and as a constituent of various metal alloys, such as sterling silver used in jewelry, cupronickel used to make marine hardware and coins, and constantan used in strain gauges and thermocouples for temperature measurement.
Copper is one of the few metals that occur in nature in directly usable metallic form as opposed to needing extraction from an ore. This led to very early human use, from c. 8000 BC. It was the first metal to be smelted from its ore, c. 5000 BC, the first metal to be cast into a shape in a mold, c. 4000 BC and the first metal to be purposefully alloyed with another metal, tin, to create bronze, c. 3500 BC.[1]
In the Roman era, copper was principally mined on Cyprus, the origin of the name of the metal, from aes сyprium (metal of Cyprus), later corrupted to сuprum, from which the words copper (English), cuivre (French), Koper (Dutch) and Kupfer (German) are all derived.[2] The commonly encountered compounds are copper(II) salts, which often impart blue or green colors to such minerals as azurite, malachite, and turquoise, and have been used widely and historically as pigments. Copper used in buildings, usually for roofing, oxidizes to form a green verdigris (or patina). Copper is sometimes used in decorative art, both in its elemental metal form and in compounds as pigments. Copper compounds are used as bacteriostatic agents, fungicides, and wood preservatives.
Copper is essential to all living organisms as a trace dietary mineral because it is a key constituent of the respiratory enzyme complex cytochrome c oxidase. In molluscs and crustaceans, copper is a constituent of the blood pigment hemocyanin, replaced by the iron-complexed hemoglobin in fish and other vertebrates. In humans, copper is found mainly in the liver, muscle, and bone.[3] The adult body contains between 1.4 and 2.1 mg of copper per kilogram of body weight.[4]
Characteristics
[sunting | sunting sumber]Physical
[sunting | sunting sumber]

Copper, silver, and gold are in group 11 of the periodic table; these three metals have one s-orbital electron on top of a filled d-electron shell and are characterized by high ductility, and electrical and thermal conductivity. The filled d-shells in these elements contribute little to interatomic interactions, which are dominated by the s-electrons through metallic bonds. Unlike metals with incomplete d-shells, metallic bonds in copper are lacking a covalent character and are relatively weak. This observation explains the low hardness and high ductility of single crystals of copper.[5] At the macroscopic scale, introduction of extended defects to the crystal lattice, such as grain boundaries, hinders flow of the material under applied stress, thereby increasing its hardness. For this reason, copper is usually supplied in a fine-grained polycrystalline form, which has greater strength than monocrystalline forms.[6]
The softness of copper partly explains its high electrical conductivity (59.6×106 S/m) and high thermal conductivity, second highest (second only to silver) among pure metals at room temperature.[7] This is because the resistivity to electron transport in metals at room temperature originates primarily from scattering of electrons on thermal vibrations of the lattice, which are relatively weak in a soft metal.[5] The maximum permissible current density of copper in open air is approximately 3.1×106 A/m2 of cross-sectional area, above which it begins to heat excessively.[8]
Copper is one of a few metallic elements with a natural color other than gray or silver.[9] Pure copper is orange-red and acquires a reddish tarnish when exposed to air. The characteristic color of copper results from the electronic transitions between the filled 3d and half-empty 4s atomic shells – the energy difference between these shells corresponds to orange light.
As with other metals, if copper is put in contact with another metal, galvanic corrosion will occur.[10]
Chemical
[sunting | sunting sumber]

Copper does not react with water, but it does slowly react with atmospheric oxygen to form a layer of brown-black copper oxide which, unlike the rust that forms on iron in moist air, protects the underlying metal from further corrosion (passivation). A green layer of verdigris (copper carbonate) can often be seen on old copper structures, such as the roofing of many older buildings[11] and the Statue of Liberty.[12] Copper tarnishes when exposed to some sulfur compounds, with which it reacts to form various copper sulfides.[13]
Isotopes
[sunting | sunting sumber]There are 29 isotopes of copper. 63Cu and 65Cu are stable, with 63Cu comprising approximately 69% of naturally occurring copper; both have a spin of 3⁄2.[14] The other isotopes are radioactive, with the most stable being 67Cu with a half-life of 61.83 hours.[14] Seven metastable isotopes have been characterized; 68mCu is the longest-lived with a half-life of 3.8 minutes. Isotopes with a mass number above 64 decay by β−, whereas those with a mass number below 64 decay by β+. 64Cu, which has a half-life of 12.7 hours, decays both ways.[15]
62Cu and 64Cu have significant applications. 62Cu is used in 62Cu-PTSM as a radioactive tracer for positron emission tomography.[16]
Occurrence
[sunting | sunting sumber]Copper is produced in massive stars[17] and is present in the Earth's crust in a proportion of about 50 parts per million (ppm).[18] It occurs as native copper, in the copper sulfides chalcopyrite and chalcocite, in the copper carbonates azurite and malachite, and in the copper(I) oxide mineral cuprite.[7] The largest mass of elemental copper discovered weighed 420 tonnes and was found in 1857 on the Keweenaw Peninsula in Michigan, US.[18] Native copper is a polycrystal, with the largest single crystal ever described measuring 4.4×3.2×3.2 cm.[19]
Production
[sunting | sunting sumber]
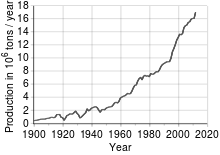

Most copper is mined or extracted as copper sulfides from large open pit mines in porphyry copper deposits that contain 0.4 to 1.0% copper. Sites include Chuquicamata in Chile, Bingham Canyon Mine in Utah, United States and El Chino Mine in New Mexico, United States. According to the British Geological Survey in 2005, Chile was the top producer of copper with at least one-third world share followed by the United States, Indonesia and Peru.[7] Copper can also be recovered through the in-situ leach process. Several sites in the state of Arizona are considered prime candidates for this method.[20] The amount of copper in use is increasing and the quantity available is barely sufficient to allow all countries to reach developed world levels of usage.[21]
Reserves
[sunting | sunting sumber]Copper has been in use at least 10,000 years, but more than 95% of all copper ever mined and smelted has been extracted since 1900,[22] and more than half was extracted the last 24 years. As with many natural resources, the total amount of copper on Earth is vast, with around 1014 tons in the top kilometer of Earth's crust, which is about 5 million years' worth at the current rate of extraction. However, only a tiny fraction of these reserves is economically viable with present-day prices and technologies. Estimates of copper reserves available for mining vary from 25 years to 60 years, depending on core assumptions such as the growth rate.[23] Recycling is a major source of copper in the modern world.[22] Because of these and other factors, the future of copper production and supply is the subject of much debate, including the concept of peak copper, analogous to peak oil.
The price of copper has historically been unstable,[24] and it sextupled from the 60-year low of US$0.60/lb (US$1.32/kg) in June 1999 to US$3.75 per pound (US$8.27/kg) in May 2006. It dropped to US$2.40/lb (US$5.29/kg) in February 2007, then rebounded to US$3.50/lb (US$7.71/kg) in April 2007.[25][butuh sumber yang lebih baik] In February 2009, weakening global demand and a steep fall in commodity prices since the previous year's highs left copper prices at US$1.51/lb (US$3.32/kg).[26]
Methods
[sunting | sunting sumber]
The concentration of copper in ores averages only 0.6%, and most commercial ores are sulfides, especially chalcopyrite (CuFeS2) and to a lesser extent chalcocite (Cu2S).[27] These minerals are concentrated from crushed ores to the level of 10–15% copper by froth flotation or bioleaching.[28] Heating this material with silica in flash smelting removes much of the iron as slag. The process exploits the greater ease of converting iron sulfides into oxides, which in turn react with the silica to form the silicate slag that floats on top of the heated mass. The resulting copper matte, consisting of Cu2S, is roasted to convert all sulfides into oxides:[27]
- 2 Cu2S + 3 O2 → 2 Cu2O + 2 SO2
The cuprous oxide is converted to blister copper upon heating:
- 2 Cu2O → 4 Cu + O2
The Sudbury matte process converted only half the sulfide to oxide and then used this oxide to remove the rest of the sulfur as oxide. It was then electrolytically refined and the anode mud exploited for the platinum and gold it contained. This step exploits the relatively easy reduction of copper oxides to copper metal. Natural gas is blown across the blister to remove most of the remaining oxygen and electrorefining is performed on the resulting material to produce pure copper:[29]
- Cu2+ + 2 e− → Cu
- Blister copper
- Smelting
- Reverberatory furnace
- Slag removal
- Copper casting of anodes
- Casting wheel
- Anodes removal machine
- Anodes take-off
- Rail cars
- Transportation to the tank house

Recycling
[sunting | sunting sumber]Like aluminium,[30] copper is recyclable without any loss of quality, both from raw state and from manufactured products.[31] In volume, copper is the third most recycled metal after iron and aluminium.[32] An estimated 80% of all copper ever mined is still in use today.[33] According to the International Resource Panel's Metal Stocks in Society report, the global per capita stock of copper in use in society is 35–55 kg. Much of this is in more-developed countries (140–300 kg per capita) rather than less-developed countries (30–40 kg per capita).
The process of recycling copper is roughly the same as is used to extract copper but requires fewer steps. High-purity scrap copper is melted in a furnace and then reduced and cast into billets and ingots; lower-purity scrap is refined by electroplating in a bath of sulfuric acid.[34]
Alloys
[sunting | sunting sumber]Numerous copper alloys have been formulated, many with important uses. Brass is an alloy of copper and zinc. Bronze usually refers to copper-tin alloys, but can refer to any alloy of copper such as aluminium bronze. Copper is one of the most important constituents of silver and carat gold and carat solders used in the jewelry industry, modifying the color, hardness and melting point of the resulting alloys.[35] Some lead-free solders consist of tin alloyed with a small proportion of copper and other metals.[36]
The alloy of copper and nickel, called cupronickel, is used in low-denomination coins, often for the outer cladding. The US 5-cent coin (currently called a nickel) consists of 75% copper and 25% nickel in homogeneous composition. The alloy of 90% copper and 10% nickel, remarkable for its resistance to corrosion, is used for various objects exposed to seawater, though it is vulnerable to the sulfides sometimes found in polluted harbors and estuaries.[37] Alloys of copper with aluminium (about 7%) have a golden color and are used in decorations.[18] Shakudō is a Japanese decorative alloy of copper containing a low percentage of gold, typically 4–10%, that can be patinated to a dark blue or black color.[38]
Compounds
[sunting | sunting sumber]
Copper forms a rich variety of compounds, usually with oxidation states +1 and +2, which are often called cuprous and cupric, respectively.[39]
Binary compounds
[sunting | sunting sumber]As with other elements, the simplest compounds of copper are binary compounds, i.e. those containing only two elements, the principal examples being oxides, sulfides, and halides. Both cuprous and cupric oxides are known. Among the numerous copper sulfides, important examples include copper(I) sulfide and copper(II) sulfide.
Cuprous halides (with chlorine, bromine, and iodine) are known, as are cupric halides with fluorine, chlorine, and bromine. Attempts to prepare copper(II) iodide yield only cuprous iodide and iodine.[39]
- 2 Cu2+ + 4 I− → 2 CuI + I2
Coordination chemistry
[sunting | sunting sumber]
Copper forms coordination complexes with ligands. In aqueous solution, copper(II) exists as [Cu(H2O)6]2+. This complex exhibits the fastest water exchange rate (speed of water ligands attaching and detaching) for any transition metal aquo complex. Adding aqueous sodium hydroxide causes the precipitation of light blue solid copper(II) hydroxide. A simplified equation is:
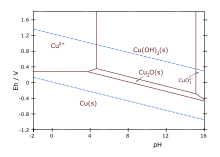
- Cu2+ + 2 OH− → Cu(OH)2
Aqueous ammonia results in the same precipitate. Upon adding excess ammonia, the precipitate dissolves, forming tetraamminecopper(II):
- Cu(H2O)4(OH)2 + 4 NH3 → [Cu(H2O)2(NH3)4]2+ + 2 H2O + 2 OH−
Many other oxyanions form complexes; these include copper(II) acetate, copper(II) nitrate, and copper(II) carbonate. Copper(II) sulfate forms a blue crystalline pentahydrate, the most familiar copper compound in the laboratory. It is used in a fungicide called the Bordeaux mixture.[40]

Polyols, compounds containing more than one alcohol functional group, generally interact with cupric salts. For example, copper salts are used to test for reducing sugars. Specifically, using Benedict's reagent and Fehling's solution the presence of the sugar is signaled by a color change from blue Cu(II) to reddish copper(I) oxide.[41] Schweizer's reagent and related complexes with ethylenediamine and other amines dissolve cellulose.[42] Amino acids form very stable chelate complexes with copper(II). Many wet-chemical tests for copper ions exist, one involving potassium ferrocyanide, which gives a brown precipitate with copper(II) salts.
Organocopper chemistry
[sunting | sunting sumber]Compounds that contain a carbon-copper bond are known as organocopper compounds. They are very reactive towards oxygen to form copper(I) oxide and have many uses in chemistry. They are synthesized by treating copper(I) compounds with Grignard reagents, terminal alkynes or organolithium reagents;[43] in particular, the last reaction described produces a Gilman reagent. These can undergo substitution with alkyl halides to form coupling products; as such, they are important in the field of organic synthesis. Copper(I) acetylide is highly shock-sensitive but is an intermediate in reactions such as the Cadiot-Chodkiewicz coupling[44] and the Sonogashira coupling.[45] Conjugate addition to enones[46] and carbocupration of alkynes[47] can also be achieved with organocopper compounds. Copper(I) forms a variety of weak complexes with alkenes and carbon monoxide, especially in the presence of amine ligands.[48]
Copper(III) and copper(IV)
[sunting | sunting sumber]Copper(III) is most often found in oxides. A simple example is potassium cuprate, KCuO2, a blue-black solid.[49] The most extensively studied copper(III) compounds are the cuprate superconductors. Yttrium barium copper oxide (YBa2Cu3O7) consists of both Cu(II) and Cu(III) centres. Like oxide, fluoride is a highly basic anion[50] and is known to stabilize metal ions in high oxidation states. Both copper(III) and even copper(IV) fluorides are known, K3CuF6 and Cs2CuF6, respectively.[39]
Some copper proteins form oxo complexes, which also feature copper(III).[51] With tetrapeptides, purple-colored copper(III) complexes are stabilized by the deprotonated amide ligands.[52]
Complexes of copper(III) are also found as intermediates in reactions of organocopper compounds.[53]
History
[sunting | sunting sumber]Copper Age
[sunting | sunting sumber]


Copper occurs naturally as native metallic copper and was known to some of the oldest civilizations on record. The history of copper use dates to 9000 BC in the Middle East;[54] a copper pendant was found in northern Iraq that dates to 8700 BC.[55] Evidence suggests that gold and meteoric iron (but not iron smelting) were the only metals used by humans before copper.[56] The history of copper metallurgy is thought to follow this sequence: First, cold working of native copper, then annealing, smelting, and, finally, lost-wax casting. In southeastern Anatolia, all four of these techniques appear more or less simultaneously at the beginning of the Neolithic c. 7500 BC.[57]
Copper smelting was independently invented in different places. It was probably discovered in China before 2800 BC, in Central America around 600 AD, and in West Africa about the 9th or 10th century AD.[58] Investment casting was invented in 4500–4000 BC in Southeast Asia[54] and carbon dating has established mining at Alderley Edge in Cheshire, UK, at 2280 to 1890 BC.[59] Ötzi the Iceman, a male dated from 3300–3200 BC, was found with an axe with a copper head 99.7% pure; high levels of arsenic in his hair suggest an involvement in copper smelting.[60] Experience with copper has assisted the development of other metals; in particular, copper smelting led to the discovery of iron smelting.[60] Production in the Old Copper Complex in Michigan and Wisconsin is dated between 6000 and 3000 BC.[61][62] Natural bronze, a type of copper made from ores rich in silicon, arsenic, and (rarely) tin, came into general use in the Balkans around 5500 BC.[butuh rujukan]
Bronze Age
[sunting | sunting sumber]Alloying copper with tin to make bronze was first practiced about 4000 years after the discovery of copper smelting, and about 2000 years after "natural bronze" had come into general use.[butuh rujukan] Bronze artifacts from the Vinča culture date to 4500 BC.[63] Sumerian and Egyptian artifacts of copper and bronze alloys date to 3000 BC.[64] The Bronze Age began in Southeastern Europe around 3700–3300 BC, in Northwestern Europe about 2500 BC. It ended with the beginning of the Iron Age, 2000–1000 BC in the Near East, and 600 BC in Northern Europe. The transition between the Neolithic period and the Bronze Age was formerly termed the Chalcolithic period (copper-stone), when copper tools were used with stone tools. The term has gradually fallen out of favor because in some parts of the world, the Chalcolithic and Neolithic are coterminous at both ends. Brass, an alloy of copper and zinc, is of much more recent origin. It was known to the Greeks, but became a significant supplement to bronze during the Roman Empire.[64]
Antiquity and Middle Ages
[sunting | sunting sumber]

In Greece, copper was known by the name chalkos (χαλκός). It was an important resource for the Romans, Greeks and other ancient peoples. In Roman times, it was known as aes Cyprium, aes being the generic Latin term for copper alloys and Cyprium from Cyprus, where much copper was mined. The phrase was simplified to cuprum, hence the English copper. Aphrodite (Venus in Rome) represented copper in mythology and alchemy because of its lustrous beauty and its ancient use in producing mirrors; Cyprus was sacred to the goddess. The seven heavenly bodies known to the ancients were associated with the seven metals known in antiquity, and Venus was assigned to copper.[65]
Copper was first used in ancient Britain in about the 3rd or 2nd Century BC. In North America, copper mining began with marginal workings by Native Americans. Native copper is known to have been extracted from sites on Isle Royale with primitive stone tools between 800 and 1600.[66] Copper metallurgy was flourishing in South America, particularly in Peru around 1000 AD. Copper burial ornamentals from the 15th century have been uncovered, but the metal's commercial production did not start until the early 20th century.
The cultural role of copper has been important, particularly in currency. Romans in the 6th through 3rd centuries BC used copper lumps as money. At first, the copper itself was valued, but gradually the shape and look of the copper became more important. Julius Caesar had his own coins made from brass, while Octavianus Augustus Caesar's coins were made from Cu-Pb-Sn alloys. With an estimated annual output of around 15,000 t, Roman copper mining and smelting activities reached a scale unsurpassed until the time of the Industrial Revolution; the provinces most intensely mined were those of Hispania, Cyprus and in Central Europe.[67][68]
The gates of the Temple of Jerusalem used Corinthian bronze treated with depletion gilding.[butuh klarifikasi][butuh rujukan] The process was most prevalent in Alexandria, where alchemy is thought to have begun.[69] In ancient India, copper was used in the holistic medical science Ayurveda for surgical instruments and other medical equipment. Ancient Egyptians (~2400 BC) used copper for sterilizing wounds and drinking water, and later to treat headaches, burns, and itching.
Modern period
[sunting | sunting sumber]
The Great Copper Mountain was a mine in Falun, Sweden, that operated from the 10th century to 1992. It satisfied two thirds of Europe's copper consumption in the 17th century and helped fund many of Sweden's wars during that time.[70] It was referred to as the nation's treasury; Sweden had a copper backed currency.[71]
Copper is used in roofing,[11] currency, and for photographic technology known as the daguerreotype. Copper was used in Renaissance sculpture, and was used to construct the Statue of Liberty; copper continues to be used in construction of various types. Copper plating and copper sheathing were widely used to protect the under-water hulls of ships, a technique pioneered by the British Admiralty in the 18th century.[72] The Norddeutsche Affinerie in Hamburg was the first modern electroplating plant, starting its production in 1876.[73] The German scientist Gottfried Osann invented powder metallurgy in 1830 while determining the metal's atomic mass; around then it was discovered that the amount and type of alloying element (e.g., tin) to copper would affect bell tones. Flash smelting was developed by Outokumpu in Finland and first applied at Harjavalta in 1949; the energy-efficient process accounts for 50% of the world's primary copper production.[74]
The Intergovernmental Council of Copper Exporting Countries, formed in 1967 by Chile, Peru, Zaire and Zambia, operated in the copper market as OPEC does in oil, though it never achieved the same influence, particularly because the second-largest producer, the United States, was never a member; it was dissolved in 1988.[75]
Applications
[sunting | sunting sumber]
The major applications of copper are electrical wire (60%), roofing and plumbing (20%), and industrial machinery (15%). Copper is used mostly as a pure metal, but when greater hardness is required, it is put into such alloys as brass and bronze (5% of total use).[18] For more than two centuries, copper paint has been used on boat hulls to control the growth of plants and shellfish.[76] A small part of the copper supply is used for nutritional supplements and fungicides in agriculture.[40][77] Machining of copper is possible, although alloys are preferred for good machinability in creating intricate parts.
Wire and cable
[sunting | sunting sumber]Despite competition from other materials, copper remains the preferred electrical conductor in nearly all categories of electrical wiring except overhead electric power transmission where aluminium is often preferred.[78][79] Copper wire is used in power generation, power transmission, power distribution, telecommunications, electronics circuitry, and countless types of electrical equipment.[80] Electrical wiring is the most important market for the copper industry.[81] This includes structural power wiring, power distribution cable, appliance wire, communications cable, automotive wire and cable, and magnet wire. Roughly half of all copper mined is used for electrical wire and cable conductors.[82] Many electrical devices rely on copper wiring because of its multitude of inherent beneficial properties, such as its high electrical conductivity, tensile strength, ductility, creep (deformation) resistance, corrosion resistance, low thermal expansion, high thermal conductivity, ease of soldering, malleability, and ease of installation.
For a short period from the late 1960s to the late 1970s, copper wiring was replaced by aluminium wiring in many housing construction projects in America. The new wiring was implicated in a number of house fires and the industry returned to copper.[83][84]
Electronics and related devices
[sunting | sunting sumber]
Integrated circuits and printed circuit boards increasingly feature copper in place of aluminium because of its superior electrical conductivity; heat sinks and heat exchangers use copper because of its superior heat dissipation properties. Electromagnets, vacuum tubes, cathode ray tubes, and magnetrons in microwave ovens use copper, as do waveguides for microwave radiation.[85]
Electric motors
[sunting | sunting sumber]Copper's superior conductivity enhances the efficiency of electrical motors.[86] This is important because motors and motor-driven systems account for 43%–46% of all global electricity consumption and 69% of all electricity used by industry.[87] Increasing the mass and cross section of copper in a coil increases the efficiency of the motor. Copper motor rotors, a new technology designed for motor applications where energy savings are prime design objectives,[88][89] are enabling general-purpose induction motors to meet and exceed National Electrical Manufacturers Association (NEMA) premium efficiency standards.[90]
Architecture
[sunting | sunting sumber]

Copper has been used since ancient times as a durable, corrosion resistant, and weatherproof architectural material.[91][92][93][94] Roofs, flashings, rain gutters, downspouts, domes, spires, vaults, and doors have been made from copper for hundreds or thousands of years. Copper's architectural use has been expanded in modern times to include interior and exterior wall cladding, building expansion joints, radio frequency shielding, and antimicrobial and decorative indoor products such as attractive handrails, bathroom fixtures, and counter tops. Some of copper's other important benefits as an architectural material include low thermal movement, light weight, lightning protection, and recyclability.
The metal's distinctive natural green patina has long been coveted by architects and designers. The final patina is a particularly durable layer that is highly resistant to atmospheric corrosion, thereby protecting the underlying metal against further weathering.[95][96][97] It can be a mixture of carbonate and sulfate compounds in various amounts, depending upon environmental conditions such as sulfur-containing acid rain.[98][99][100][101] Architectural copper and its alloys can also be 'finished' to embark a particular look, feel, and/or color. Finishes include mechanical surface treatments, chemical coloring, and coatings.[102]
Copper has excellent brazing and soldering properties and can be welded; the best results are obtained with gas metal arc welding.[103]
Antibiofouling applications
[sunting | sunting sumber]Copper is biostatic, meaning bacteria and many other forms of life will not grow on it. For this reason it has long been used to line parts of ships to protect against barnacles and mussels. It was originally used pure, but has since been superseded by Muntz metal and copper-based paint. Similarly, as discussed in copper alloys in aquaculture, copper alloys have become important netting materials in the aquaculture industry because they are antimicrobial and prevent biofouling, even in extreme conditions[104] and have strong structural and corrosion-resistant[105] properties in marine environments.
Antimicrobial applications
[sunting | sunting sumber]Copper-alloy touch surfaces have natural properties that destroy a wide range of microorganisms (e.g., E. coli O157:H7, methicillin-resistant Staphylococcus aureus (MRSA), Staphylococcus, Clostridium difficile, influenza A virus, adenovirus, and fungi).[106] Some 355 copper alloys[butuh klarifikasi] were proven to kill more than 99.9% of disease-causing bacteria within just two hours when cleaned regularly.[107] The United States Environmental Protection Agency (EPA) has approved the registrations of these copper alloys as "antimicrobial materials with public health benefits";[107] that approval allows manufacturers to make legal claims to the public health benefits of products made of registered alloys. In addition, the EPA has approved a long list of antimicrobial copper products made from these alloys, such as bedrails, handrails, over-bed tables, sinks, faucets, door knobs, toilet hardware, computer keyboards, health club equipment, and shopping cart handles (for a comprehensive list, see: Antimicrobial copper-alloy touch surfaces#Approved products). Copper doorknobs are used by hospitals to reduce the transfer of disease, and Legionnaires' disease is suppressed by copper tubing in plumbing systems.[108] Antimicrobial copper alloy products are now being installed in healthcare facilities in the U.K., Ireland, Japan, Korea, France, Denmark, and Brazil[butuh rujukan] and in the subway transit system in Santiago, Chile, where copper-zinc alloy handrails will be installed in some 30 stations between 2011 and 2014.[109][110][111]
Folk medicine
[sunting | sunting sumber]Copper is commonly used in jewelry, and according to some folklore, copper bracelets relieve arthritis symptoms.[112] In various studies, though, no difference is found between arthritis treated with a copper bracelet, magnetic bracelet, or placebo bracelet.[113][114] Medical science has not demonstrated any benefits in copper jewelry for any medical condition. A human being can have a dietary copper deficiency, but the condition is very rare because copper is present in many common foods, including legumes (beans), grains, and nuts.[115]
No evidence shows that copper can be absorbed through the skin. If it were, it might lead to copper poisoning.[116]
Compression clothing
[sunting | sunting sumber]Recently, some compression clothing with inter-woven copper has been marketed with the same folk medicine claims. Because compression clothing is a valid treatment for some ailments, the clothing may appear to work, but the added copper may have no benefit beyond a placebo effect.[117]
Other uses
[sunting | sunting sumber]Templat:Repetition section Solutions of copper compounds are used as a wood preservative, particularly in treating the original portion of structures during restoration of dry rot damage. Together with zinc, copper wires may be installed over non-conductive roofing materials to discourage the growth of moss.[butuh rujukan] Textile fibers are blended with copper to create antimicrobial protective fabrics.[118][119] Copper alloys are used in musical instruments, particularly: the body of brass instruments; circuitry for all those that are electronically amplified; the bodies of brass percussion such as gongs, bells, and kettle drums; tuning heads on guitars and other string instruments; string windings on harps, pianos, harpsichords, and string instruments; and the frame elements of pianos and harps. Copper is commonly used as a base on which other metals such as nickel are electroplated.
Copper is one of three metals, along with lead and silver, used in the museum materials testing procedure called the Oddy test to detect chlorides, oxides, and sulfur compounds.
Copper is used as the printing plate in etching, engraving and other forms of intaglio printmaking.
Copper oxide and carbonate are used add color in stain glass works, in glassmaking, and in ceramic glazes to impart turquoise blue, green, and brown colors.
Copper is used to create stills for distilling spirits, for example to make whisky. It's malleability makes it easy to bend into the various shapes required and allows considerable flexibility in the shaping of the still and associated pipework; the metal also reacts with undesirable sulfur-containing components in the vapor and distillate making for a cleaner product.
Degradation
[sunting | sunting sumber]Chromobacterium violaceum and Pseudomonas fluorescens can both mobilize solid copper as a cyanide compound.[120] The ericoid mycorrhizal fungi associated with Calluna, Erica and Vaccinium can grow in metalliferous soils containing copper.[120] The ectomycorrhizal fungus Suillus luteus protects young pine trees from copper toxicity. A sample of the fungus Aspergillus niger was found growing from gold mining solution and was found to contain cyano complexes of such metals as gold, silver, copper, iron, and zinc. The fungus also plays a role in the solubilization of heavy metal sulfides.[121]
Biological role
[sunting | sunting sumber]
Copper proteins have diverse roles in biological electron transport and oxygen transportation, processes that exploit the easy interconversion of Cu(I) and Cu(II).[122][123][124] [125] The biological role for copper commenced with the appearance of oxygen in earth's atmosphere.[126]
Copper is essential in the aerobic respiration of all eukaryotes. In mitochondria, it is found in cytochrome c oxidase, which is the last protein in oxidative phosphorylation. Cytochrome c oxidase is the protein that binds the O2 between a copper and an iron; the protein transfers 8 electrons to the O2 molecule to reduce it to two molecules of water.
Copper is also found in many superoxide dismutases, proteins that catalyze the decomposition of superoxides by converting it (by disproportionation) to oxygen and hydrogen peroxide:
- 2 HO2 → H2O2 + O2
The protein hemocyanin is the oxygen carrier in most mollusks and some arthropods such as the horseshoe crab (Limulus polyphemus).[127] Because hemocyanin is blue, these organisms have blue blood rather than the red blood of iron-based hemoglobin. Structurally related to hemocyanin are the laccases and tyrosinases. Instead of reversibly binding oxygen, these proteins hydroxylate substrates, illustrated by their role in the formation of lacquers.[125]
Several copper proteins, such as the "blue copper proteins", do not interact directly with substrates, hence they are not enzymes. These proteins relay electrons by the process called electron transfer.[125]

A unique tetranuclear copper center has been found in nitrous-oxide reductase.[128]
Dietary needs
[sunting | sunting sumber]Copper is an essential trace element in plants and animals, but not all microorganisms. The human body contains copper at a level of about 1.4 to 2.1 mg per kg of body mass.[129] Copper is absorbed in the gut, then transported to the liver bound to albumin.[130] After processing in the liver, copper is distributed to other tissues in a second phase, which involves the protein ceruloplasmin, carrying the majority of copper in blood. Ceruloplasmin also carries the copper that is excreted in milk, and is particularly well-absorbed as a copper source.[131] Copper in the body normally undergoes enterohepatic circulation (about 5 mg a day, vs. about 1 mg per day absorbed in the diet and excreted from the body), and the body is able to excrete some excess copper, if needed, via bile, which carries some copper out of the liver that is not then reabsorbed by the intestine.[132][133]
Dietary reference intake
[sunting | sunting sumber]The Food and Nutrition Board of the U.S. Institute of Medicine updated the estimated average requirements (EARs) and recommended dietary allowances (RDAs) for copper in 2001. The current EAR for copper for people age 14 and older is 0.7 mg/day. The RDA is 0.9 mg/day. RDAs are higher than EARs so as to identify amounts that will cover people with higher than average requirements. The RDA for pregnant women equals 1.0 mg/day. The RDA for lactating women equals 1.3 mg/day. For infants up to 12 months the AI is 0.22 mg/day and for children age 1–13 years the RDA increases with age from 0.34 to 0.7 mg/day. The Food and Nutrition Board also sets tolerable upper intake Levels (ULs) for vitamins and minerals when evidence is sufficient. In the case of copper the UL is set at 10 mg/day. Collectively the EARs, RDAs, AIs and ULs are referred to as Dietary Reference Intakes.[134] The European Food Safety Authority reviewed the same safety question and set its UL at 5 mg/day.[135]
For U.S. food and dietary supplement labeling purposes the amount in a serving is expressed as a percent of daily value (%DV). For copper labeling purposes 100% of the DV was 2.0 mg, but as of May 2016 it has been revised to 0.9 mg. Food and supplement companies have until July 28, 2018 to comply with the change.
Copper-based disorders
[sunting | sunting sumber]Because of its role in facilitating iron uptake, copper deficiency can produce anemia-like symptoms, neutropenia, bone abnormalities, hypopigmentation, impaired growth, increased incidence of infections, osteoporosis, hyperthyroidism, and abnormalities in glucose and cholesterol metabolism. Conversely, Wilson's disease causes an accumulation of copper in body tissues.
Severe deficiency can be found by testing for low plasma or serum copper levels, low ceruloplasmin, and low red blood cell superoxide dismutase levels; these are not sensitive to marginal copper status. The "cytochrome c oxidase activity of leucocytes and platelets" has been stated as another factor in deficiency, but the results have not been confirmed by replication.[136]
Gram quantities of various copper salts have been taken in suicide attempts and produced acute copper toxicity in humans, possibly due to redox cycling and the generation of reactive oxygen species that damage DNA.[137][138] Corresponding amounts of copper salts (30 mg/kg) are toxic in animals.[139] A minimum dietary value for healthy growth in rabbits has been reported to be at least 3 ppm in the diet.[140] However, higher concentrations of copper (100 ppm, 200 ppm, or 500 ppm) in the diet of rabbits may favorably influence feed conversion efficiency, growth rates, and carcass dressing percentages.[141]
Chronic copper toxicity does not normally occur in humans because of transport systems that regulate absorption and excretion. Autosomal recessive mutations in copper transport proteins can disable these systems, leading to Wilson's disease with copper accumulation and cirrhosis of the liver in persons who have inherited two defective genes.[129]
Elevated copper levels have also been linked to worsening symptoms of Alzheimer's disease.[142][143]
Human exposure
[sunting | sunting sumber]In the US, the Occupational Safety and Health Administration (OSHA) has designated a permissible exposure limit (PEL) for copper dust and fumes in the workplace as a time-weighted average (TWA) of 1 mg/m3. The National Institute for Occupational Safety and Health (NIOSH) has set a Recommended exposure limit (REL) of 1 mg/m3, time-weighted average. The IDLH (immediately dangerous to life and health) value is 100 mg/m3.[144]
Copper is a constituent of tobacco smoke.[145][146] The tobacco plant readily absorbs and accumulates heavy metals, such as copper from the surrounding soil into its leaves. These are readily absorbed into the user's body following smoke inhalation.[147] The health implications are not clear.[148]
See also
[sunting | sunting sumber]- Erosion corrosion of copper water tubes
- List of countries by copper production
- Metal theft
- Smelter
- Peak copper
References
[sunting | sunting sumber]- ^ McHenry, Charles, ed. (1992). The New Encyclopedia Britannica. 3 (edisi ke-15). Chicago: Encyclopedia Britannica, Inc. hlm. 612. ISBN 0-85229-553-7.
- ^ Encyclopaedia Britannica, 11th ed., vol. 7, p. 102.
- ^ Johnson, MD PhD, Larry E., ed. (2008). "Copper". Merck Manual Home Health Handbook. Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. Diakses tanggal 7 April 2013.
- ^ "Copper in human health".
- ^ a b George L. Trigg; Edmund H. Immergut (1 November 1992). Encyclopedia of applied physics. 4: Combustion to Diamagnetism. VCH Publishers. hlm. 267–272. ISBN 978-3-527-28126-8. Diakses tanggal 2 May 2011.
- ^ Smith, William F. & Hashemi, Javad (2003). Foundations of Materials Science and Engineering. McGraw-Hill Professional. hlm. 223. ISBN 0-07-292194-3.
- ^ a b c Hammond, C. R. (2004). The Elements, in Handbook of Chemistry and Physics (edisi ke-81st). CRC press. ISBN 0-8493-0485-7.
- ^ Resistance Welding Manufacturing Alliance (2003). Resistance Welding Manual (edisi ke-4th). Resistance Welding Manufacturing Alliance. hlm. 18–12. ISBN 0-9624382-0-0.
- ^ Chambers, William; Chambers, Robert (1884). Chambers's Information for the People. L (edisi ke-5th). W. & R. Chambers. hlm. 312. ISBN 0-665-46912-8.
- ^ "Galvanic Corrosion". Corrosion Doctors. Diakses tanggal 29 April 2011.
- ^ a b Grieken, Rene van; Janssens, Koen (2005-09-27). Cultural Heritage Conservation and Environmental Impact Assessment by Non-Destructive Testing and Micro-Analysis (dalam bahasa Inggris). CRC Press. hlm. 197. ISBN 978-0-203-97078-2.
- ^ "Copper.org: Education: Statue of Liberty: Reclothing the First Lady of Metals – Repair Concerns". Copper.org. Diakses tanggal 11 April 2011.
- ^ Rickett, B. I.; Payer, J. H. (1995). "Composition of Copper Tarnish Products Formed in Moist Air with Trace Levels of Pollutant Gas: Hydrogen Sulfide and Sulfur Dioxide/Hydrogen Sulfide". Journal of the Electrochemical Society. 142 (11): 3723–3728. doi:10.1149/1.2048404.
- ^ a b Audi, G; Bersillon, O.; Blachot, J.; Wapstra, A. H. (2003). "Nubase2003 Evaluation of Nuclear and Decay Properties". Nuclear Physics A. Atomic Mass Data Center. 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001.
- ^ "Interactive Chart of Nuclides". National Nuclear Data Center. Diakses tanggal 8 April 2011.
- ^ Okazawad, Hidehiko; Yonekura, Yoshiharu; Fujibayashi, Yasuhisa; Nishizawa, Sadahiko; Magata, Yasuhiro; Ishizu, Koichi; Tanaka, Fumiko; Tsuchida, Tatsuro; Tamaki, Nagara; Konishi, Junji (1994). "Clinical Application and Quantitative Evaluation of Generator-Produced Copper-62-PTSM as a Brain Perfusion Tracer for PET" (PDF). Journal of Nuclear Medicine. 35 (12): 1910–1915. PMID 7989968.
- ^ Romano, Donatella; Matteucci, Fransesca (2007). "Contrasting copper evolution in ω Centauri and the Milky Way". Monthly Notices of the Royal Astronomical Society: Letters. 378 (1): L59–L63. arXiv:astro-ph/0703760
 . Bibcode:2007MNRAS.378L..59R. doi:10.1111/j.1745-3933.2007.00320.x.
. Bibcode:2007MNRAS.378L..59R. doi:10.1111/j.1745-3933.2007.00320.x.
- ^ a b c d Emsley, John (11 August 2003). Nature's building blocks: an A-Z guide to the elements. Oxford University Press. hlm. 121–125. ISBN 978-0-19-850340-8. Diakses tanggal 2 May 2011.
- ^ Rickwood, P. C. (1981). "The largest crystals" (PDF). American Mineralogist. 66: 885.
- ^ Randazzo, Ryan (19 June 2011). "A new method to harvest copper". Azcentral.com. Diakses tanggal 25 April 2014.
- ^ Gordon, R. B.; Bertram, M.; Graedel, T. E. (2006). "Metal stocks and sustainability". Proceedings of the National Academy of Sciences. 103 (5): 1209–1214. Bibcode:2006PNAS..103.1209G. doi:10.1073/pnas.0509498103. PMC 1360560
 . PMID 16432205.
. PMID 16432205.
- ^ a b Leonard, Andrew (2 March 2006). "Peak copper?". Salon – How the World Works. Diarsipkan dari versi asli tanggal 7 March 2008. Diakses tanggal 23 March 2008.
- ^ Brown, Lester (2006). Plan B 2.0: Rescuing a Planet Under Stress and a Civilization in Trouble. New York: W.W. Norton. hlm. 109. ISBN 0-393-32831-7.
- ^ Schmitz, Christopher (1986). "The Rise of Big Business in the World, Copper Industry 1870–1930". Economic History Review. 2. 39 (3): 392–410. doi:10.1111/j.1468-0289.1986.tb00411.x. JSTOR 2596347.
- ^ "Copper Trends: Live Metal Spot Prices".
- ^ Ackerman, R. (2 April 2009). "A Bottom In Sight For Copper". Forbes.
- ^ a b Greenwood, Norman N.; Earnshaw, A. (1997), Chemistry of the Elements (edisi ke-2), Oxford: Butterworth-Heinemann, ISBN 0-7506-3365-4
- ^ Watling, H. R. (2006). "The bioleaching of sulphide minerals with emphasis on copper sulphides — A review" (PDF). Hydrometallurgy. 84 (1, 2): 81–108. doi:10.1016/j.hydromet.2006.05.001. Diarsipkan dari versi asli (PDF) tanggal 18 August 2011.
- ^ Samans, Carl (1949). Engineering metals and their alloys. New York: Macmillan. OCLC 716492542.
- ^ Burton, Julie McCulloch (2015-02-20). Pen to Paper: Making Fun of Life (dalam bahasa Inggris). iUniverse. ISBN 9781491753941.
- ^ Bahadir, Ali Mufit; Duca, Gheorghe (2009-08-03). The Role of Ecological Chemistry in Pollution Research and Sustainable Development (dalam bahasa Inggris). Springer. ISBN 9789048129034.
- ^ Green, Dan (2016-09-06). The Periodic Table in Minutes (dalam bahasa Inggris). Quercus. ISBN 9781681443294.
- ^ "International Copper Association".
- ^ "Overview of Recycled Copper" Copper.org. Copper.org (25 August 2010). Retrieved on 8 November 2011.
- ^ "Gold Jewellery Alloys". World Gold Council. Diarsipkan dari versi asli tanggal 14 April 2009. Diakses tanggal 6 June 2009.
- ^ Balver Zinn Solder Sn97Cu3 Diarsipkan 7 July 2011 di Wayback Machine.. (PDF) . balverzinn.com. Retrieved on 8 November 2011.
- ^ Corrosion Tests and Standards (dalam bahasa Inggris). ASTM International. hlm. 368.
- ^ Oguchi, Hachiro (1983). "Japanese Shakudō: its history, properties and production from gold-containing alloys". Gold Bulletin. World Gold Council. 16 (4): 125–132. doi:10.1007/BF03214636.
- ^ a b c Holleman, A. F.; Wiberg, N. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 978-0-12-352651-9.
- ^ a b Wiley-Vch (2 April 2007). "Nonsystematic (Contact) Fungicides". Ullmann's Agrochemicals. hlm. 623. ISBN 978-3-527-31604-5.
- ^ Ralph L. Shriner, Christine K. F. Hermann, Terence C. Morrill, David Y. Curtin, Reynold C. Fuson "The Systematic Identification of Organic Compounds" 8th edition, J. Wiley, Hoboken. ISBN 0-471-21503-1
- ^ Saalwächter, Kay; Burchard, Walther; Klüfers, Peter; Kettenbach, G.; Mayer, Peter; Klemm, Dieter; Dugarmaa, Saran (2000). "Cellulose Solutions in Water Containing Metal Complexes". Macromolecules. 33: 4094–4107. doi:10.1021/ma991893m.
- ^ "Modern Organocopper Chemistry" Norbert Krause, Ed., Wiley-VCH, Weinheim, 2002. ISBN 978-3-527-29773-3.
- ^ Berná, José; Goldup, Stephen; Lee, Ai-Lan; Leigh, David; Symes, Mark; Teobaldi, Gilberto; Zerbetto, Fransesco (26 May 2008). "Cadiot–Chodkiewicz Active Template Synthesis of Rotaxanes and Switchable Molecular Shuttles with Weak Intercomponent Interactions". Angewandte Chemie. 120 (23): 4464–4468. doi:10.1002/ange.200800891.
- ^ Rafael Chinchilla & Carmen Nájera (2007). "The Sonogashira Reaction: A Booming Methodology in Synthetic Organic Chemistry". Chemical Reviews. 107 (3): 874–922. doi:10.1021/cr050992x. PMID 17305399.
- ^ "An Addition of an Ethylcopper Complex to 1-Octyne: (E)-5-Ethyl-1,4-Undecadiene" (PDF). Organic Syntheses. 64: 1. 1986. doi:10.15227/orgsyn.064.0001. Diarsipkan dari versi asli (PDF) tanggal 19 June 2012.
- ^ Kharasch, M. S.; Tawney, P. O. (1941). "Factors Determining the Course and Mechanisms of Grignard Reactions. II. The Effect of Metallic Compounds on the Reaction between Isophorone and Methylmagnesium Bromide". Journal of the American Chemical Society. 63 (9): 2308–2316. doi:10.1021/ja01854a005.
- ^ Imai, Sadako; Fujisawa, Kiyoshi; Kobayashi, Takako; Shirasawa, Nobuhiko; Fujii, Hiroshi; Yoshimura, Tetsuhiko; Kitajima, Nobumasa; Moro-oka, Yoshihiko (1998). "63Cu NMR Study of Copper(I) Carbonyl Complexes with Various Hydrotris(pyrazolyl)borates: Correlation between 63Cu Chemical Shifts and CO Stretching Vibrations". Inorganic Chemistry. 37 (12): 3066–3070. doi:10.1021/ic970138r.
- ^ G. Brauer, ed. (1963). "Potassium Cuprate (III)". Handbook of Preparative Inorganic Chemistry. 1 (edisi ke-2nd). NY: Academic Press. hlm. 1015.
- ^ Schwesinger, Reinhard; Link, Reinhard; Wenzl, Peter; Kossek, Sebastian (2006). "Anhydrous phosphazenium fluorides as sources for extremely reactive fluoride ions in solution". Chemistry: A European Journal. 12 (2): 438–45. doi:10.1002/chem.200500838. PMID 16196062.
- ^ Lewis, E. A.; Tolman, W. B. (2004). "Reactivity of Dioxygen-Copper Systems". Chemical Reviews. 104 (2): 1047–1076. doi:10.1021/cr020633r. PMID 14871149.
- ^ McDonald, M. R.; Fredericks, F. C.; Margerum, D. W. (1997). "Characterization of Copper(III)-Tetrapeptide Complexes with Histidine as the Third Residue". Inorganic Chemistry. 36 (14): 3119–3124. doi:10.1021/ic9608713. PMID 11669966.
- ^ Greenwood, Norman N.; Earnshaw, A. (1997), Chemistry of the Elements (edisi ke-2), Oxford: Butterworth-Heinemann, hlm. 1187, ISBN 0-7506-3365-4
- ^ a b "CSA – Discovery Guides, A Brief History of Copper". Csa.com. Diakses tanggal 12 September 2008.
- ^ Rayner W. Hesse (2007). Jewelrymaking through History: an Encyclopedia. Greenwood Publishing Group. hlm. 56. ISBN 0-313-33507-9.No primary source is given in that book.
- ^ "Copper". Elements.vanderkrogt.net. Diakses tanggal 12 September 2008.
- ^ Renfrew, Colin (1990). Before civilization: the radiocarbon revolution and prehistoric Europe. Penguin. ISBN 978-0-14-013642-5. Diakses tanggal 21 December 2011.
- ^ Cowen, R. "Essays on Geology, History, and People: Chapter 3: Fire and Metals". Diakses tanggal 7 July 2009.
- ^ Timberlake, S. & Prag A.J.N.W. (2005). The Archaeology of Alderley Edge: Survey, excavation and experiment in an ancient mining landscape. Oxford: John and Erica Hedges Ltd. hlm. 396.
- ^ a b "CSA – Discovery Guides, A Brief History of Copper". CSA Discovery Guides. Diakses tanggal 29 April 2011.
- ^ Pleger, Thomas C. "A Brief Introduction to the Old Copper Complex of the Western Great Lakes: 4000–1000 BC", Proceedings of the Twenty-seventh Annual Meeting of the Forest History Association of Wisconsin, Oconto, Wisconsin, 5 October 2002, pp. 10–18.
- ^ Emerson, Thomas E. and McElrath, Dale L. Archaic Societies: Diversity and Complexity Across the Midcontinent, SUNY Press, 2009 ISBN 1-4384-2701-8.
- ^ Radivojević, Miljana; Rehren, Thilo (December 2013). "Tainted ores and the rise of tin bronzes in Eurasia, c. 6500 years ago". Antiquity Publications Ltd.
- ^ a b McNeil, Ian (2002). Encyclopaedia of the History of Technology. London ; New York: Routledge. hlm. 13, 48–66. ISBN 0-203-19211-7.
- ^ Rickard, T. A. (1932). "The Nomenclature of Copper and its Alloys". Journal of the Royal Anthropological Institute. Royal Anthropological Institute. 62: 281. doi:10.2307/2843960. JSTOR 2843960.
- ^ Martin, Susan R. (1995). "The State of Our Knowledge About Ancient Copper Mining in Michigan". The Michigan Archaeologist. 41 (2–3): 119.
- ^ Hong, S.; Candelone, J.-P.; Patterson, C. C.; Boutron, C. F. (1996). "History of Ancient Copper Smelting Pollution During Roman and Medieval Times Recorded in Greenland Ice". Science. 272 (5259): 246–249 (247f.). Bibcode:1996Sci...272..246H. doi:10.1126/science.272.5259.246.
- ^ de Callataÿ, François (2005). "The Graeco-Roman Economy in the Super Long-Run: Lead, Copper, and Shipwrecks". Journal of Roman Archaeology. 18: 361–372 (366–369).
- ^ Savenije, Tom J.; Warman, John M.; Barentsen, Helma M.; van Dijk, Marinus; Zuilhof, Han; Sudhölter, Ernst J. R. (2000). "Corinthian Bronze and the Gold of the Alchemists" (PDF). Macromolecules. 33 (2): 60–66. Bibcode:2000MaMol..33...60S. doi:10.1021/ma9904870. Diarsipkan dari versi asli (PDF) tanggal 29 September 2007.
- ^ Lynch, Martin (15 April 2004). Mining in World History. hlm. 60. ISBN 978-1-86189-173-0.
- ^ "Gold: prices, facts, figures and research: A brief history of money". Diakses tanggal 22 April 2011.
- ^ "Copper and Brass in Ships". Diakses tanggal 6 September 2016.
- ^ Stelter, M.; Bombach, H. (2004). "Process Optimization in Copper Electrorefining". Advanced Engineering Materials. 6 (7): 558–562. doi:10.1002/adem.200400403.
- ^ "Outokumpu Flash Smelting" (PDF). Outokumpu. hlm. 2. Diarsipkan dari versi asli (PDF) tanggal 24 July 2011.
- ^ Karen A. Mingst (1976). "Cooperation or illusion: an examination of the intergovernmental council of copper exporting countries". International Organization. 30 (2): 263–287. doi:10.1017/S0020818300018270.
- ^ Ryck Lydecker. "Is Copper Bottom Paint Sinking?". BoatUS Magazine. Diakses tanggal 2016-06-03.
- ^ "Copper". American Elements. 2008. Diakses tanggal 12 July 2008.
- ^ Pops, Horace, 2008, "Processing of wire from antiquity to the future", Wire Journal International, June, pp. 58–66
- ^ The Metallurgy of Copper Wire, http://www.litz-wire.com/pdf%20files/Metallurgy_Copper_Wire.pdf
- ^ Joseph, Günter, 1999, Copper: Its Trade, Manufacture, Use, and Environmental Status, edited by Kundig, Konrad J.A., ASM International, pps. 141–192 and pps. 331–375.
- ^ "Copper, Chemical Element – Overview, Discovery and naming, Physical properties, Chemical properties, Occurrence in nature, Isotopes". Chemistryexplained.com. Diakses tanggal 16 October 2012.
- ^ Joseph, Günter, 1999, Copper: Its Trade, Manufacture, Use, and Environmental Status, edited by Kundig, Konrad J.A., ASM International, p.348
- ^ "Aluminum Wiring Hazards and Pre-Purchase Inspections". www.heimer.com. Diakses tanggal 2016-06-03.
- ^ "Electrical Wiring FAQ (Part 2 of 2)Section - Aluminum wiring". www.faqs.org. Diakses tanggal 2016-06-03.
- ^ "Accelerator: Waveguides (SLAC VVC)". SLAC Virtual Visitor Center. Diakses tanggal 29 April 2011.
- ^ IE3 energy-saving motors, Engineer Live, http://www.engineerlive.com/Design-Engineer/Motors_and_Drives/IE3_energy-saving_motors/22687/
- ^ Energy‐efficiency policy opportunities for electric motor‐driven systems, International Energy Agency, 2011 Working Paper in the Energy Efficiency Series, by Paul Waide and Conrad U. Brunner, OECD/IEA 2011
- ^ Fuchsloch, J. and E.F. Brush, (2007), "Systematic Design Approach for a New Series of Ultra‐NEMA Premium Copper Rotor Motors", in EEMODS 2007 Conference Proceedings, 10–15 June, Beijing.
- ^ Copper motor rotor project; Copper Development Association; "Archived copy". Diarsipkan dari versi asli tanggal 13 March 2012. Diakses tanggal 2012-11-07.
- ^ NEMA Premium Motors, The Association of Electrical Equipment and Medical Imaging Manufacturers; "Archived copy". Diarsipkan dari versi asli tanggal 2 April 2010. Diakses tanggal 2009-10-12.
- ^ Seale, Wayne (2007). The role of copper, brass, and bronze in architecture and design; Metal Architecture, May 2007
- ^ Copper roofing in detail; Copper in Architecture; Copper Development Association, U.K., www.cda.org.uk/arch
- ^ Architecture, European Copper Institute; http://eurocopper.org/copper/copper-architecture.html
- ^ Kronborg completed; Agency for Palaces and Cultural Properties, København, "Archived copy". Diarsipkan dari versi asli tanggal 24 October 2012. Diakses tanggal 2012-09-12.
- ^ Berg, Jan. "Why did we paint the library's roof?". Diarsipkan dari versi asli tanggal 25 June 2007. Diakses tanggal 20 September 2007.
- ^ Architectural considerations; Copper in Architecture Design Handbook, http://www.copper.org/applications/architecture/arch_dhb/fundamentals/arch_considerations.htm
- ^ Peters, Larry E. (2004). Preventing corrosion on copper roofing systems; Professional Roofing, October 2004, http://www.professionalroofing.net
- ^ Oxidation Reaction: Why is the Statue of Liberty Blue-Green? Engage Students in Engineering; www.EngageEngineering.org; Chun Wu, Ph.D., Mount Marty College; Funded by the National Science Foundation (NSF) under Grant No. 083306. "Archived copy" (PDF). Diarsipkan dari versi asli (PDF) tanggal 25 October 2013. Diakses tanggal 2013-10-25.
- ^ Fitzgerald, K. P.; Nairn, J.; Atrens, A. (1998). "The chemistry of copper patination". Corrosion Science. 40 (12): 2029–50. doi:10.1016/S0010-938X(98)00093-6.
- ^ Application Areas: Architecture – Finishes – patina; http://www.copper.org/applications/architecture/finishes.html
- ^ Glossary of copper terms, Copper Development Association (UK): "Archived copy". Diarsipkan dari versi asli tanggal 20 August 2012. Diakses tanggal 2012-09-14.
- ^ Finishes – natural weathering; Copper in Architecture Design Handbook, Copper Development Association Inc., "Archived copy". Diarsipkan dari versi asli tanggal 16 October 2012. Diakses tanggal 2012-09-12.
- ^ Davis, Joseph R. (2001). Copper and Copper Alloys. ASM International. hlm. 3–6, 266. ISBN 0-87170-726-8.
- ^ Edding, Mario E., Flores, Hector, and Miranda, Claudio, (1995), Experimental Usage of Copper-Nickel Alloy Mesh in Mariculture. Part 1: Feasibility of usage in a temperate zone; Part 2: Demonstration of usage in a cold zone; Final report to the International Copper Association Ltd.
- ^ Corrosion Behaviour of Copper Alloys used in Marine Aquaculture. (PDF) . copper.org. Retrieved on 8 November 2011.
- ^ Copper Touch Surfaces. Copper Touch Surfaces. Retrieved on 8 November 2011.
- ^ a b EPA registers copper-containing alloy products, May 2008
- ^ Biurrun, Amaya; Caballero, Luis; Pelaz, Carmen; León, Elena; Gago, Alberto (1999). "Treatment of a Legionella pneumophila‐Colonized Water Distribution System Using Copper‐Silver Ionization and Continuous Chlorination". Infection Control and Hospital Epidemiology. 20 (6): 426–428. doi:10.1086/501645. JSTOR 30141645. PMID 10395146.
- ^ Chilean subway protected with Antimicrobial Copper – Rail News from Diarsipkan 24 July 2012 di Wayback Machine.. rail.co. Retrieved on 8 November 2011.
- ^ Codelco to provide antimicrobial copper for new metro lines (Chile) [pranala nonaktif]. Construpages.com.ve. Retrieved on 8 November 2011.
- ^ PR 811 Chilean Subway Installs Antimicrobial Copper Diarsipkan 23 November 2011 di Wayback Machine.. (PDF). antimicrobialcopper.com. Retrieved on 8 November 2011.
- ^ Walker, W. R.; Keats, D. M. (1976). "An investigation of the therapeutic value of the 'copper bracelet'-dermal assimilation of copper in arthritic/rheumatoid conditions". Agents and actions. 6 (4): 454–9. PMID 961545.
- ^ Richmond, SJ; Brown, SR; Campion, PD; et al. (2009). "Therapeutic effects of magnetic and copper bracelets in osteoarthritis: a randomised placebo-controlled crossover trial". Complement Ther Med. 17: 249–56. doi:10.1016/j.ctim.2009.07.002. PMID 19942103.
No difference was observed between devices in terms of their effects on pain as measured by the primary outcome measure (WOMAC A), the PRI and the VAS. Similar results were obtained for stiffness (WOMAC B), physical function (WOMAC C), and medication use. Further analyses of the PRI subscales revealed a statistically significant difference between devices (P=0.025), which favoured the experimental device. Participants reported lower sensory pain after wearing the standard magnetic wrist strap, than when wearing control devices. However, no adjustment was made for multiple testing. - ^ Richmond, Stewart J.; Brown, Sally R.; Campion, Peter D.; Porter, Amanda J. L.; Moffett, Jennifer A. Klaber; Jackson, David A.; Featherstone, Valerie A.; Taylor, Andrew J. (2009). "Therapeutic effects of magnetic and copper bracelets in osteoarthritis: A randomised placebo-controlled crossover trial". Complementary Therapies in Medicine. 17 (5–6): 249–256. doi:10.1016/j.ctim.2009.07.002. ISSN 0965-2299. PMID 19942103.
- ^ University of Arkansas for Medical Sciences:
Can wearing a copper bracelet cure arthritis?
According to the Center for Hand and Upper Extremity Surgery at UAMS, copper deficiency is extremely rare and most regular diets provide enough copper to meet the daily requirements. Copper is a component of some of the normal cellular enzymes in most mineral rich foods, such as vegetables, potatoes, legumes (beans and peas), nuts (peanuts and pecans), grains (wheat and rye) and fruits. Supplementation is only needed in patients with serious medical conditions that affect their gastrointestinal tract and impair their ability to absorb nutrients. - ^ University of Arkansas for Medical Sciences:
Find the Truth Behind Medical Myths Diarsipkan 6 January 2014 di Wayback Machine.
While it's never been proven that copper can copper be absorbed through the skin by wearing a bracelet, research has shown that excessive copper can result in poisoning, causing vomiting and, in severe cases, liver damage. - ^
Truth in Advertising
Tommie Copper
So it seems possible that copper-infused compression clothing could help you recover from a tough workout, and it's also possible it could have some anti-bacterial properties in clothes. But as for the claims in the infomercial about relieving joint pain and helping with everyday aches — any relief from copper-compression seems more likely to be a placebo effect than anything else. Think carefully before shelling out for Tommie Copper. - ^ "Copper and Cupron". Cupron. [sumber tepercaya?]
- ^ Ergowear, Copper antimicrobial yarn technology used in male underwear [sumber tepercaya?]
- ^ a b Geoffrey Michael Gadd (March 2010). "Metals, minerals and microbes: geomicrobiology and bioremediation". Microbiology. 156 (3): 609–643. doi:10.1099/mic.0.037143-0. PMID 20019082.
- ^ Harbhajan Singh (2006-11-17). Mycoremediation: Fungal Bioremediation. hlm. 509. ISBN 978-0-470-05058-3.
- ^ Vest, Katherine E.; Hashemi, Hayaa F.; Cobine, Paul A. (2013). "Chapter 13 The Copper Metallome in Eukaryotic Cells". Dalam Banci, Lucia. Metallomics and the Cell. Metal Ions in Life Sciences. 12. Springer. doi:10.1007/978-94-007-5561-10_12. ISBN 978-94-007-5560-4. electronic-book ISBN 978-94-007-5561-1 ISSN 1559-0836 electronic-ISSN 1868-0402
- ^ Vest, Katherine E.; Hashemi, Hayaa F.; Cobine, Paul A. (2013). "Chapter 12 The Copper Metallome in Prokaryotic Cells". Dalam Banci, Lucia. Metallomics and the Cell. Metal Ions in Life Sciences. 12. Springer. doi:10.1007/978-94-007-5561-10_13. ISBN 978-94-007-5560-4. electronic-book ISBN 978-94-007-5561-1 ISSN 1559-0836 electronic-ISSN 1868-0402
- ^ Yee, Gereon M.; Tolman, William B. (2015). "Chapter 5, Section 4 Dioxygen Activation by Copper Complexes". Dalam Peter M.H. Kroneck and Martha E. Sosa Torres. Sustaining Life on Planet Earth: Metalloenzymes Mastering Dioxygen and Other Chewy Gases. Metal Ions in Life Sciences. 15. Springer. hlm. 175–192. doi:10.1007/978-3-319-12415-5_5.
- ^ a b c S. J. Lippard, J. M. Berg "Principles of bioinorganic chemistry" University Science Books: Mill Valley, CA; 1994. ISBN 0-935702-73-3.
- ^ Decker, H. & Terwilliger, N. (2000). "COPs and Robbers: Putative evolution of copper oxygen-binding proteins". Journal of Experimental Biology. 203 (Pt 12): 1777–1782. PMID 10821735.
- ^ "Fun facts". Horseshoe crab. University of Delaware. Diakses tanggal 13 July 2008.
- ^ Schneider, Lisa K.; Wüst, Anja; Pomowski, Anja; Zhang, Lin; Einsle, Oliver (2014). "Chapter 8. No Laughing Matter: The Unmaking of the Greenhouse Gas Dinitrogen Monoxide by Nitrous Oxide Reductase". Dalam Peter M.H. Kroneck; Martha E. Sosa Torres. The Metal-Driven Biogeochemistry of Gaseous Compounds in the Environment. Metal Ions in Life Sciences. 14. Springer. hlm. 177–210. doi:10.1007/978-94-017-9269-1_8.
- ^ a b "Amount of copper in the normal human body, and other nutritional copper facts". Diakses tanggal 3 April 2009.
- ^ Adelstein, S. J.; Vallee, B. L. (1961). "Copper metabolism in man". New England Journal of Medicine. 265 (18): 892–897. doi:10.1056/NEJM196111022651806.
- ^ M C Linder; Wooten, L.; Cerveza, P.; Cotton, S.; Shulze, R.; Lomeli, N. (1 May 1998). "Copper transport". The American Journal of Clinical Nutrition. 67 (5): 965S–971S. PMID 9587137.
- ^ Frieden, E.; Hsieh, H. S. (1976). "Ceruloplasmin: The copper transport protein with essential oxidase activity". Advances in Enzymology and Related Areas of Molecular Biology. Advances in Enzymology - and Related Areas of Molecular Biology. 44: 187–236. doi:10.1002/9780470122891.ch6. ISBN 978-0-470-12289-1. JSTOR 20170553. PMID 775938.
- ^ S. S. Percival; Harris, E. D. (1 January 1990). "Copper transport from ceruloplasmin: Characterization of the cellular uptake mechanism". American Journal of Physiology. Cell Physiology. 258 (1): C140–6. PMID 2301561.
- ^ Copper. IN: Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Copper. National Academy Press. 2001, PP. 224–257.
- ^ Tolerable Upper Intake Levels For Vitamins And Minerals (PDF), European Food Safety Authority, 2006
- ^ Bonham, Maxine; O'Connor, Jacqueline M.; Hannigan, Bernadette M.; Strain, J. J. (2002). "The immune system as a physiological indicator of marginal copper status?". British Journal of Nutrition. 87 (5): 393–403. doi:10.1079/BJN2002558. PMID 12010579.
- ^ Li, Yunbo; Trush, Michael; Yager, James (1994). "DNA damage caused by reactive oxygen species originating from a copper-dependent oxidation of the 2-hydroxy catechol of estradiol". Carcinogenesis. 15 (7): 1421–1427. doi:10.1093/carcin/15.7.1421. PMID 8033320.
- ^ Gordon, Starkebaum; John, M. Harlan (April 1986). "Endothelial cell injury due to copper-catalyzed hydrogen peroxide generation from homocysteine". J. Clin. Invest. 77 (4): 1370–6. doi:10.1172/JCI112442. PMC 424498
 . PMID 3514679.
. PMID 3514679.
- ^ "Pesticide Information Profile for Copper Sulfate". Cornell University. Diakses tanggal 10 July 2008.
- ^ Hunt, Charles E. & William W. Carlton (1965). "Cardiovascular Lesions Associated with Experimental Copper Deficiency in the Rabbit". Journal of Nutrition. 87 (4): 385–394. PMID 5841854.
- ^ Ayyat M.S.; Marai I.F.M.; Alazab A.M. (1995). "Copper-Protein Nutrition of New Zealand White Rabbits under Egyptian Conditions". World Rabbit Science. 3 (3): 113–118. doi:10.4995/wrs.1995.249.
- ^ Brewer GJ. Copper excess, zinc deficiency, and cognition loss in Alzheimer's disease. BioFactors (Oxford, England). March 2012;38(2):107–113. doi:10.1002/biof.1005. PMID 22438177.
- ^ "Copper: Alzheimer's Disease". Examine.com. Diakses tanggal 21 June 2015.
- ^ "NIOSH Pocket Guide to Chemical Hazards #0150". National Institute for Occupational Safety and Health (NIOSH).
- ^ OEHHA Copper
- ^ Talhout, Reinskje; Schulz, Thomas; Florek, Ewa; Van Benthem, Jan; Wester, Piet; Opperhuizen, Antoon (2011). "Hazardous Compounds in Tobacco Smoke". International Journal of Environmental Research and Public Health. 8 (12): 613–628. doi:10.3390/ijerph8020613. ISSN 1660-4601. PMC 3084482
 . PMID 21556207.
. PMID 21556207.
- ^ Alireza Pourkhabbaz, Hamidreza Pourkhabbaz Investigation of Toxic Metals in the Tobacco of Different Iranian Cigarette Brands and Related Health Issues, Iran J Basic Med Sci. 2012 Jan-Feb; 15(1): 636–644. PMC 3586865
- ^ David Bernhard, Andrea Rossmann, and Georg Wick Metals in Cigarette Smoke, IUBMB Life, 57(12): 805–809, December 2005
Notes
[sunting | sunting sumber]| in pure water, or acidic or alkali conditions. Copper in neutral water is more noble than hydrogen. | in water containing sulfide | in 10 M ammonia solution | in a chloride solution |
Further reading
[sunting | sunting sumber]- Massaro, Edward J., ed. (2002). Handbook of Copper Pharmacology and Toxicology. Humana Press. ISBN 0-89603-943-9.
- "Copper: Technology & Competitiveness (Summary) Chapter 6: Copper Production Technology" (PDF). Office of Technology Assessment. 2005.
- Current Medicinal Chemistry, Volume 12, Number 10, May 2005, pp. 1161–1208(48) Metals, Toxicity and Oxidative Stress
- William D. Callister (2003). Materials Science and Engineering: an Introduction (edisi ke-6th). Wiley, New York. Table 6.1, p. 137. ISBN 0-471-73696-1.
- Material: Copper (Cu), bulk, MEMS and Nanotechnology Clearinghouse.
- Kim BE; Nevitt T; Thiele DJ (2008). "Mechanisms for copper acquisition, distribution and regulation". Nat. Chem. Biol. 4 (3): 176–85. doi:10.1038/nchembio.72. PMID 18277979.
- Copper transport disorders: an Instant insight from the Royal Society of Chemistry
External links
[sunting | sunting sumber]- Copper at The Periodic Table of Videos (University of Nottingham)
- Copper and compounds fact sheet from the National Pollutant Inventory of Australia
- CDC–NIOSH Pocket Guide to Chemical Hazards – Copper (dusts and mists) from the Centers for Disease Control and Prevention's National Institute for Occupational Safety and Health
- CDC–NIOSH Pocket Guide to Chemical Hazards – Copper fume from the Centers for Disease Control and Prevention's National Institute for Occupational Safety and Health
- Copper.org - official website of the Copper Development Association with an extensive site of properties and uses of copper
- Price history of copper, according to the IMF
| (besar) | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | ||||||||||||||||
| 1 | H | He | |||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | |||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | |||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | |||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | |||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
|
| |||||||||||||||||||||||||||||||||

